Tetanus
A non-infectious disease with no natural immunity1
- Toxin-mediated bacterial disease (highly potent tetanus toxin)1
- Infection through contact/reactivation of spores in tissue lesions1
- No naturally-acquired immunity: recovery from disease does not confer immunity1
- Non communicable disease2
- Infection may occur at any age (high mortality in neonate & >65 yr)1
- Elimination of environmental exposure is impossible (C. tetani spores are ubiquitous)2

- Immunity only through passive or active immunization1
- In the absence of definite correlation of protection, high anti-body concentrations should be maintained throughout life1
- WHO. WER, 2016;92(6)
- Roper. Tetanus Toxoid. In: Vaccines, 2018
Waning Anti-Tetanus Immunity
A School-entry booster helps maintain high immunity1
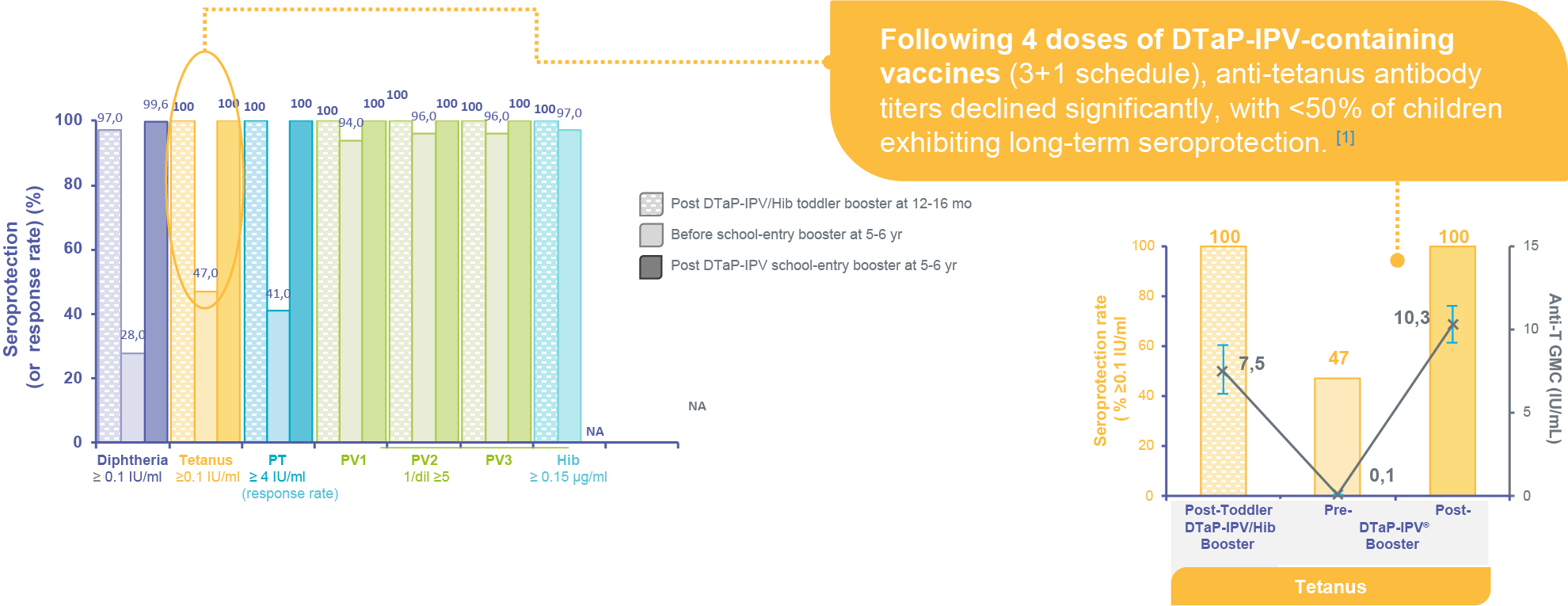
Immune responses after a DTaP-IPV/Hib toddler booster (n=69) and before/2 months after DTaP-IPV school-entry booster (n=232) at 5-6 years in children primed w/ DTaP-IPV/Hib at 2-3-4/2-4-6 months (3+1 schedule, France, 2000-01)1
NA: not available; mo: months; yr: years
- Mallet. Vaccine, 2004;22(11-12)
Importance of DTaP-IPV School-Entry Booster
Tetanus

Tetanus is a non-communicable disease with no natural immunity caused by the ubiquitous spores of Clostridium tetani1,2
Prevention through vaccination is the best option against this non eradicable disease1
- In the absence of an absolute correlate of protection, the aim of vaccination should be to maintain high antibody levels throughout life1,2
- After the 3rd dose, each additional dose increases antitoxin antibody levels and prolongs the duration of immunity1
- A school-entry booster is needed to compensate for vaccine-induced waning immunity and will provide protection into adolescence3
- WHO. WER, 2016;92(6)
- Roper. Tetanus Toxoid. In: Vaccines, 2018
- Borrow. Immunologic Basis for immunization: Module 3: Tetanus. 2007
Sustained Anti-Tetanus Immunity
Relies on primary series plus several boosters including a school-entry booster1

TTCV: tetanus toxoid-containing vaccine
- Borrow. Immunologic Basis for immunization: Module 3: Tetanus. 2007
Vaccination Recommendations - WHO
School-entry boosters are recommended for long-lived protection1-5

- WHO. WER, 2017;92(31)
- WHO. WER, 2017;92(6)
- WHO. WER, 2015;90(351)
- WHO. WER, 2016;91(12)
- Martinon-Torres. Expert Rev Vaccines, 2018;17(4)
MAT-AE-2200199-V1-March-2022