Bilateral Endoscopic Nasal Polyp Score (NPS)1,3
Polyp size is assessed endoscopically to determine the nasal polyp score (NPS).
The NPS scale ranges from 0 (no polyp) to 4 (large polyps) for each nostril. These scores are tallied from each nostril for a total score ranging from 0 to 8.
| Stage | Endoscopic Appearance | Description |
| 0 |  |
No polyps. |
| 1 |  |
Small polyps restricted to the middle meatus; do not extend below the lower border of the middle turbinate. |
| 2 |  |
Polyps extend below the lower border of the middle turbinate. |
| 3 |  |
Large polyps extend down to the lower border of the inferior turbinate, or polyps are medial to the middle turbinate. |
| 4 |  |
Large polyps that result in complete obstruction of the nasal cavity. |
22-Item Sino-Nasal Outcome Test (SNOT-22)1,6
The 22-item sino-nasal outcome test (SNOT-22) is a validated questionnaire used to assess the impact of CRSwNP on HRQoL.
It includes questions specifically designed to measure nasal blockage, and sense of taste and smell.
- The range of the SNOT-22 score is 0 to 110.
- Lower scores indicate less impact.
Take a Closer Look: The SNOT-22
The SNOT-22 questionnaire provides a 6-point scale which is used to assess 22 symptoms and social or emotional consequences in patients with CRSwNP.
Each symptom or consequence is ranked on a 6-point scale:
- No problem (0 pts)
- Very mild problem (1 pt)
- Mild or slight problem (2 pts)
- Moderate problem (3 pts)
- Severe problem (4 pts)
- Problem as bad as it can be (5 pts)
The following list contains the 22 symptoms and social or emotional consequences ranked in the SNOT-22:
- Need to blow nose
- Sneezing
- Runny nose
- Cough
- Post nasal discharge
- Thick nasal discharge
- Ear fullness
- Dizziness
- Ear pain
- Facial pain/pressure
- Difficulty falling asleep
- Waking up at night
- Lack of good night’s sleep
- Waking up tired
- Fatigue
- Reduced productivity
- Reduced concentration
- Frustrated/restless/irritable
- Sad
- Embarrassed
- Sense of taste/smell
- Blockage/congestion of nose
Lund-Mackay CT Scoring System1,4,5
The Lund-Mackay scoring system is used in conjunction with CT for scanning of nasal polyps.
| Sinus System | Possible Score* | Grading |
| Maxillary | 0, 1, or 2 | 0 = no mucosal thickening 1 = partial opacification 2 = total opacification |
| Anterior ethmoid | 0, 1, or 2 | |
| Posterior ethmoid | 0, 1, or 2 | |
| Sphenoid | 0, 1, or 2 | |
| Frontal | 0, 1, or 2 | |
| Osteomeatal complex | 0 or 2 only |
CT Computed Tomography
*Calculated per side (total score of 0 to 24 across both sinuses)
This system uses a score of 0 to 2, which reflects the absence (0), partial (1), or complete (2) opacification of each sinus as well as of the vital ostiomeatal complex (0 is not obstructed and 2 is obstructed).
The maximum score possible for each side is 12 (total score of 0 to 24 across both sides), higher scores indicate worse status.
Evaluation of Nasal Symptoms and Olfactory Function11
Nasal Congestion Score
The nasal congestion score (NCS) is a self-evaluation of symptoms of congestion.
The NCS is a monthly average of the patient’s own daily morning assessment of the severity of their congestion symptoms over the previous 24 hours.
The NCS is graded according to the following 4-point scale:
0 is equal to absent symptoms (no sign/symptom evident).
1 is equal to mild symptoms (sign/symptom clearly present, but minimal awareness; easily tolerated).
2 is equal to moderate symptoms (definite awareness of sign/symptom that is bothersome but tolerable).
3 is equal to severe symptoms (sign/symptom that is hard to tolerate; causes interference with activities of daily living and/or sleeping).
Total Rhinosinusitis Symptoms Score (TSS)1,2
The Total Rhinosinusitis Symptoms Score (TSS) is a calculated composite score (ranging between 0 to 9) consisting of the sum of rhinosinusitis symptoms assessed each morning by the patient.
TSS assesses the following symptoms:
- Nasal congestion.
- Loss of sense of smell.
- Rhinorrhea.
Each symptom is graded on a 4-point scale (0 - 3):
0 indicates no symptoms.
1 indicates mild symptoms that are easily tolerated.
2 indicates awareness of symptoms that are bothersome but tolerable.
3 indicates severe symptoms that are hard to tolerate and interfere with daily activity.
Olfactory Dysfunction13
- Loss of smell is one of the most significant symptoms to patients, and has a substantial impact on QoL.
- In addition, the impairment of the sense of smell seems to correlate with disease severity and maybe the first sign of disease recurrence.
- However, the exact mechanism underlying impaired olfaction is still not fully understood.
- May be related to nasal airflow reduction, local edema-induced compression of olfactory nerves, as well as mucosal inflammation.
Definitions of Smell Impairment14
| Term | Description |
| Normosmia | Normal sense of smell |
| Hyposmia | Decreased sense of smell |
| Hyperosmia | Increased sense of smell |
| Anosmia | Total loss of smell |
| Specific anosmia | Inability to identify a certain odor |
| Parosmia | Aberrant perception of odor |
Loss of Smell Score (LoS)1,2
The loss of smell score is a patient-collected assessment of the daily symptom severity of decreased or loss of smell.
The loss of smells score is graded using a 0 to 3 categorical scale, where 0 = no symptom, 1 = mild LoS, 2 = moderate LoS, 3 = severe LoS.
VAS score
Visual Analog Scale (VAS), 0 to 10.

This VAS is ideal in the classification of symptoms of rhinosinusitis into mild (VAS 0–3), moderate (>3–7), and severe (>7–10).
University of Pennsylvania Smell Identification Test (UPSIT)7,8
- A self-administered test that uses microencapsulated odorants that are released by scratching standardized odor-impregnated test booklets
- The individual “scratches and sniffs” each odor in the booklet, then selects from four multiple-choice answers corresponding to the perceived smell
- The UPSIT score ranges from 0 to 40, where higher scores indicate better status
A particular strength of this test is that a clinician can distinguish patients:
- Normal sense of smell, normosmia score (35-40)
- Different levels of reduction; mild (score of 31), moderate (score of 26-30)
- Severe microsmia score (9-25), anosmia score (0-18)
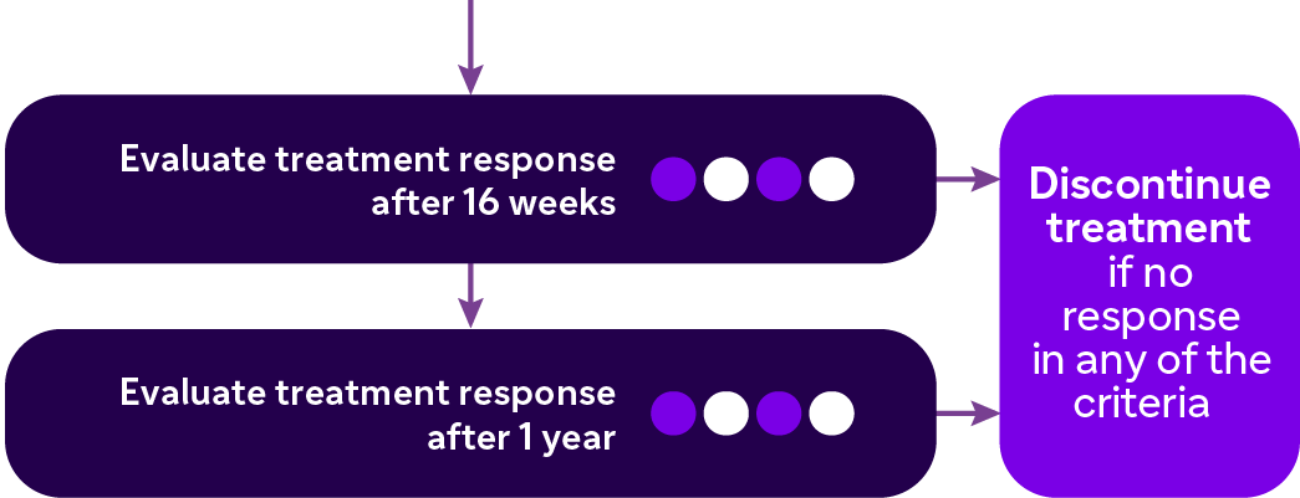
Updates in the Guidelines 20199
EUFOREA* Guidelines
Evaluation of 5 criteria
- Reduced nasal polyp size
- Reduced need for systemic corticosteroids
- Improved quality of life
- Improved sense of smell
- Reduced impact of comorbidities
Response For Treatment
EUFOREA* Consensus

- Evidence of Type 2 Inflammation
- Need for systemic corticosteroids (2 or more courses in the past year)
- Significantly impaired quality of life
- Significant loss of smell
- Diagnosis of comorbid asthma
*European Forum for Research and Education in Allergy and Airway diseases
Updates in the Guidelines 202010
EPOS** Guideline

| Criteria | Cut-off Points |
| Evidence of Type 2 Inflammation | Tissue eos ≥ 10/hpf, OR blood eos ≥ 250, OR total IgE ≥100 |
| Need for systemic corticosteroids or contraindication to systemic steroids | ≥ 2 courses per yr, OR long term (>3 months) low dose steroids |
| Significantly impaired quality of life | SNOT-22 ≥ 40 |
| Significant loss of smell | Anosmic on smell test (score depending on test) |
| Diagnosis of comorbid asthma | Asthma needing regular inhaled corticosteroids |
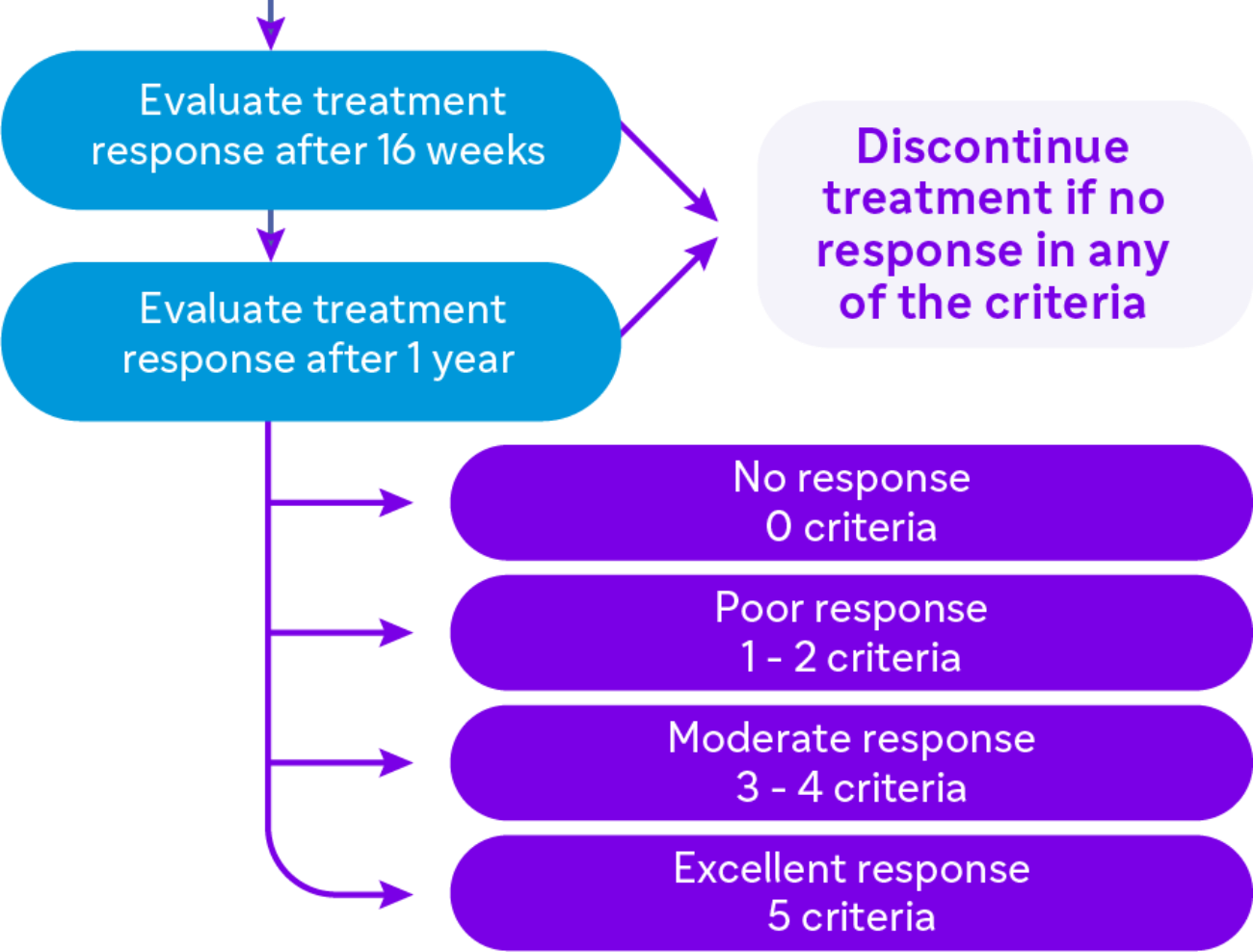
Response For Treatment
Defining response to biological treatment in CRSwNP
Evaluation of 5 criteria
- Reduced nasal polyp size
- Reduced need for systemic corticosteroids
- Improved quality of life
- Improved sense of smell
- Reduced impact of comorbidities
Excellent response
5 criteria |
Moderate response
3 - 4 criteria |
Poor response
1 - 2 criteria |
No response 0 criteria |
*Exceptional circumstances excluded (e.g., not fit for surgery)
**European Position Paper on Rhinosinusitis and Nasal Polyps
EUFOREA Expert Board Meeting on Uncontrolled Severe CRSwNP15
Evaluation of the clinical response to a biologic within 6 months of treatment: ‘‘continue or stop’’ suggestions
Selection of a biologic drug and monitoring of its effectiveness
Prediction of response in an individual patient is not possible today



Diagnosis of Uncontrolled Severe CRSwNP
Uncontrolled
Persistent or recurring CRSwNP despite long-term INCS, and having received at least one course of systemic corticosteroids* in the preceding 2 years and/or previous sinonasal surgery*
- Long-term low dose systemic corticosteroids is not recommended in CRSwNP
- One course of systemic corticosteroids refers to a minimum of 5 days of systemic corticosteroids at a dose of 0.5-1 mg/Kg/day or more
- Previous sinonasal surgery refers to any surgical procedure from the resection of polyps to conventional ESS or extended approaches
Severe
Bilaterlal CRSwNP with a NPS of ≥ 4, and persistent symptoms despite long-term INCS with the need for add-on treatment
- Bilateral polyposis (by nasal endoscopy)
- NPS ≥ 4 out of 8
- Presence of persistent symptoms assessed by:
- Loss of smell score (0-3) ≥ 2 points
- NCS (0-3) ≥ 2 points
- SNOTT-22 ≥ 35 points
- Total symptoms VAS ≥ 5 out of 10 cm
*unless having a medical contraindication/rejected by the patient
For the indication of Type 2 biologics including anti-IL4 receptor alpha (Dupilumab), anti-IgE (Omalizumab) and anti-IL5/R (Mepolizumab, Benralizumab), an underlying Type 2 inflammation should be highly likely
ESS, Endoscopic Sinus Surgery; INCS, Inhaled Corticosteroid; NCS, Nasal Congestion Score; NPS, Nasal Polyp Score; SNOT-22, 22-item Sino-Nasal Outcome Test; VAS, Visual Analog Scale.
- Bachert C, Mannent L Naclerio RM, et al. Effect of Subcutaneous Dupilumab on Nasal Polyp Burden in Patients With Chronic Sinusitis and Nasal Polyposis: A Randomized Clinical Trial. JAMA. 2016 Feb 2;315(5):469-79.
- Fokkens W, Lund V, Mullol J, et al European Position Paper on Rhinosinusitis and Nasal Polyps group. European position paper on rhinosinusitis and nasal polyps 2007. Rhinol suppl.2007;(20):1-136.
- Gevaert P, Calus L Van Zele T, et al. Omalizumab is effective in allergic and nonallergic patients with nasal polyps and asthma. J Allergy Clin Immunol. 2013;131(1):110-116
- Lund VJ, Mackay IS. Staging in rhinosinusitus. Rhinology. 1993;31(4):183-184.
- Bhattacharyya N. Test-retest reliability of CT in the assessment of chronic rhinosinusitis. Laryngoscope. 1999;109(7 Pt 1):1055-1058.
- Hopkins C, Gillett S, Slack R, et al. Psychometric validity of the 22-item Sinonasal Outcome Test. Clin Otolaryngol. 2009;34(5):447-454.
- Scadding G, Hellings P, Alobid I, et al. Diagnostic tools in Rhinology EAACI position paper. Clin Transl Allergy. 2011;1(1):2.
- Doty RL, Frye RE, Agrawal U. Internal consistency reliability of the fractionated and whole University of Pennsylvania Smell Identification Test. Precept Psychophys.
- EUFOREA expert board meeting on uncontrolled severe chronic rhinosinusitis with nasal polyps (CRSwNP) and biologics: Definitions and management, J Allergy Clin Immunol 2021 Jan;147(1):29-36. doi: 10.1016/j.jaci.2020.11.013.Epub 2020 Nov 20.
- European Position Paper on Rhinosinusitis and Nasal Polyps 2020, 2020 Feb 20;58(Suppl S29):1-464. doi: 10.4193/Rhin20.600.
- Pentewar, G.. Wagh, R.. & Chincholkar, A. (2017). Pharmacoeconomic assessment and comparing effcacy between cetirizine, levocetirizine, loratadine and fexofenadine in allergic rhinitis patients. International Journal of Basic & Clinical Pharmacology, 6(11), 2684-2689. doi:http://dx.doi.org/10.18203/2319-2003.ijbcp20174788
- Ellis et al. Allergy, Asthma & Clinical Immunology (2015) 11:16 DOI 10.1186/s13223-015-0082-0.
- Gevaert, Philippe et al. "Efficacy and safety of ornalizumab in nasal polyposis: 2 randomized phase 3 trials. " The Journal of allergy and clinical immunology vol. 146.3 (2020): 595-605.doi:10.1016/j.jaci.2020.05.032.
- Li X, Lui F. Anosmia. [Updated 2020 Jul 6]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482152/.
- Bachert, Claus et al. “EUFOREA expert board meeting on uncontrolled severe chronic rhinosinusitis with nasal polyps (CRSwNP) and biologics: Definitions and management.” The Journal of allergy and clinical immunology vol. 147,1 (2021): 29-36. doi:10.1016/j.jaci.2020.11.013.
MAT-BH-2200295/ V1/MARCH2022