The Bright Study

The first head-to-head RCT designed to compare the efficacy and safety of Gla-300 with IDeg-100 in participants with T2DM.
Background

However, direct clinical comparisons between these two-2nd generation basal insulin analogues are not available.
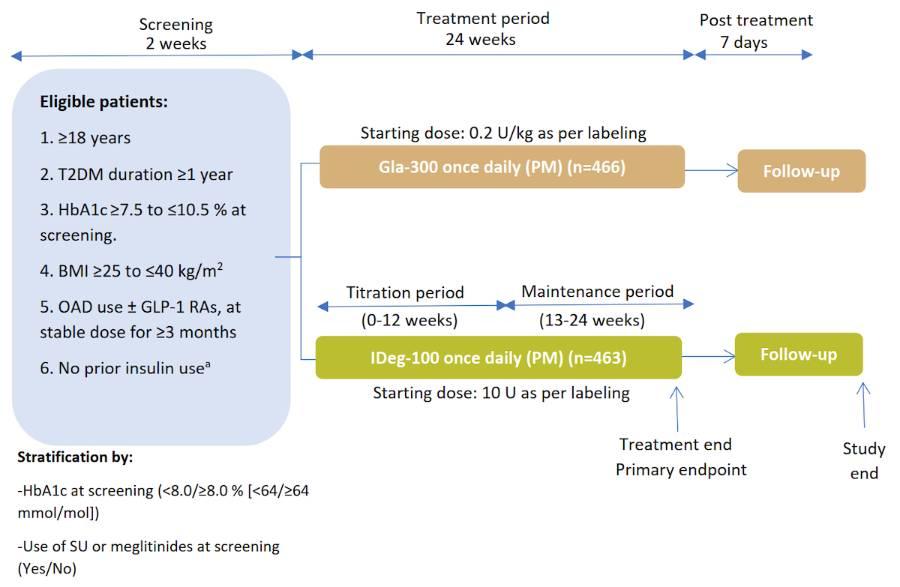
Study Design⁵
Multicentre, open-label, 1:1 randomised, active-controlled, 2-arm parallel-group, non-inferiority study in adult participants with uncontrolled T2DM
%20(1).jpg)
Gla-300 and IDeg-100 were self-administered once daily between 18:00–20:00 h5
Titrated weekly to target fasting SMPG of 4.4–5.6 mmol/L (80–100 mg/dL)
without hypoglycaemia5
Titration performed with aim of target achievement within 8–12 weeks post
randomisation (titration period)5
ᵃDoses titrated at least weekly
aWith the exception of a maximum of 8 consecutive days or 15 days total prior insulin use
Pre-Defined Endpoints⁵
Primary efficacy endpoint:
Change in HbA1c from baseline to week 24
- Analysed using a MMRM approach, adjusted for covariates including baseline HbA1c
Secondary efficacy endpoints:
-
Change in HbA1c and fasting SMPG from baseline to week 12
-
Change in FPG, fasting SMPG and 8-point SMPG profiles from baseline to week 24
-
Variability of 8-point SMPG profiles
Safety endpoints:
-
Incidence and annualised rates of confirmed hypoglycaemia (≤3.9 and <3.0 mmol/L) over the full 24-week period, and during weeks 0–12 (titration period) and weeks 13–24 (maintenance period)
-
TEAEs
Results
Primary efficacy endpoints:
Non-inferiority of Gla-300 vs IDeg-100 in HbA1c reduction at study end⁵
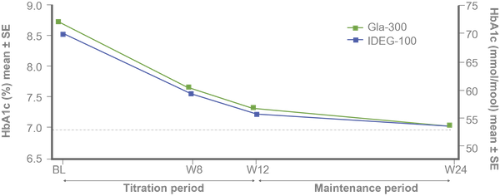
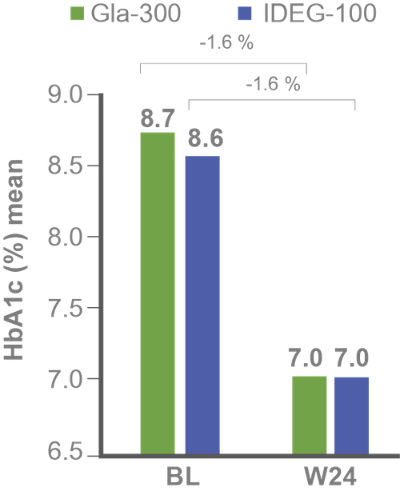
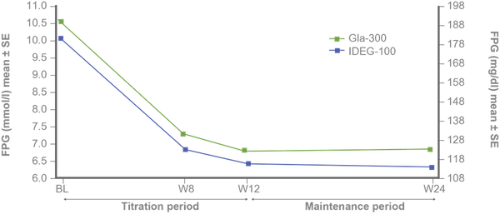
Secondary efficacy endpoints:
FPG and fasting SMPG reduction with Gla-300 vs IDeg-100 from baseline to study end⁵
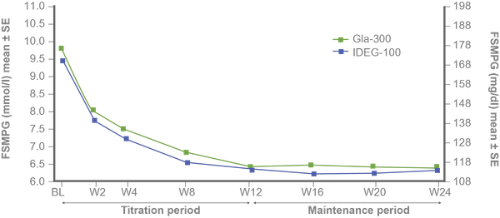
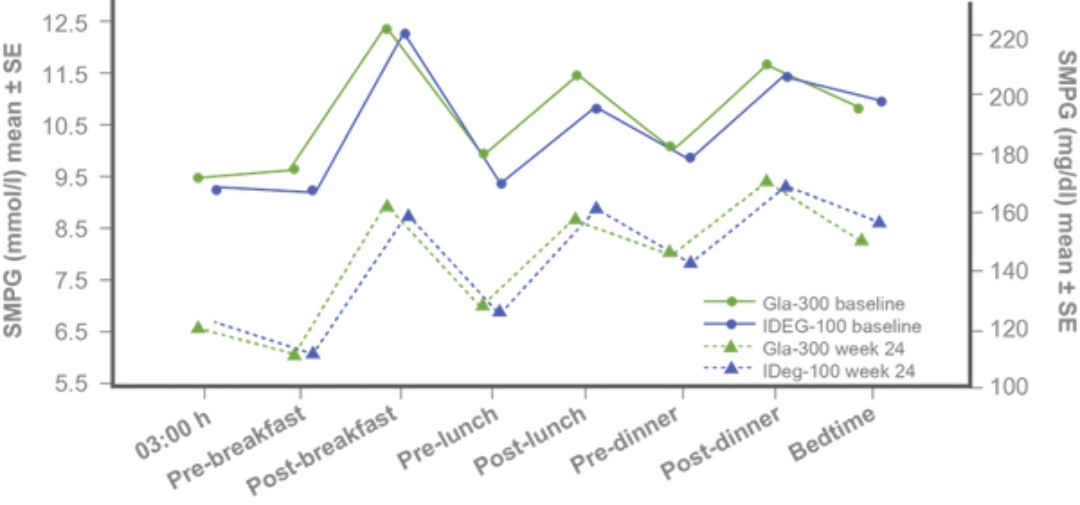
Similar 8-point SMPG and variability profiles at baseline and study end⁵
Similar variability of 24-h SMPG and fasting SMPG at baseline and week 24 with both treatments.⁵
| Mean CV, % | Gla-300 | IDeg-100 |
| 24-h SMPG |
|
|
| Baseline | 22.5 | 23.4 |
| Week 24 | 27.6 | 28.0 |
| LS mean change | 3.7 | 4.0 |
| Fasting SMPG |
|
|
| Baseline | 13.8 | 14.6 |
| Week 24 | 16.5 | 17.0 |
| LS mean change | 1.5 | 2.0 |
Safety endpoints:
Incidence (%) and events per patient-year of confirmed (≤3.9 mmol/l) during titration period⁵

Treatment-emergent adverse events⁵
-
No specific safety concerns were reported
-
There was one death in the Gla-300 group (adenocarcinoma of the colon)
-
Only one episode of severe hypoglycemia occurred during the entire study.
Confirmed hypoglycaemia included documented symptomatic or asymptomatic hypoglycaemia (≤70 mg/dL or <54 mg/dL), and severe events if any; only 1 participant experienced severe hypoglycaemia (1 event), in the Gla-300 group, due to a skipped evening meal and not reducing her insulin dose after a non-severe event 2 days earlier. All p-values presented are nominal. Safety population (Gla-300, n=463; IDeg-100, n=462)⁵
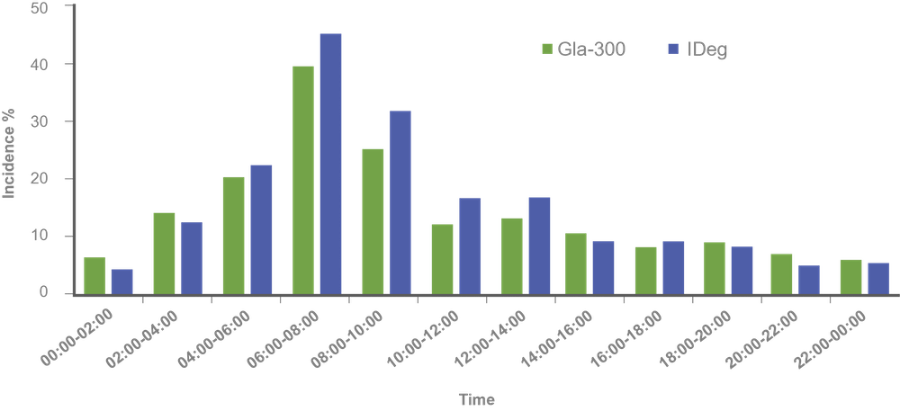
Findings:
Hypoglycemia incidence by time of day⁷
Comparable glycaemic control and hypoglycaemia incidence over 24 weeks, regardless of SU/glinide use at screening⁶
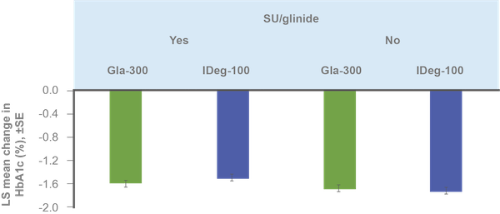
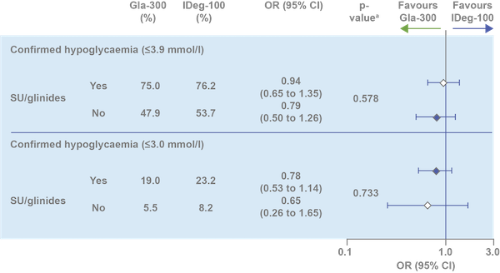
ᵃp value to test heterogeneity of treatment-by-subgroup interaction. HbA1c reduction assessed in ITT population; hypoglycemia incidence assessed in safety population

Clinical Implications
-
Gla-300 and IDeg-100 provide similar glycaemic control accompanied by comparable hypoglycaemia⁵
-
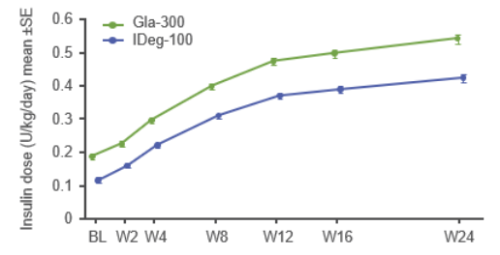
Active and adequate titration can help patients achieve glycaemic goals – dose increases are associated with improved HbA1c levels⁸
-
Early hypoglycaemic events during titration are associated with increased rates of treatment discontinuation, as well as long-term risk of hypoglycaemia⁹,¹⁰
-
Gla-300 may offer an advantage in hypoglycaemia risk reduction vs IDeg-100 during the titration period in insulin-naïve patients with T2DM⁵
Conclusion
-
Bright was the first direct comparison of Gla-300 vs IDeg-100:
-
Similar glycaemic control for HbA1c and fasting SMPG
-
Similar variability in 24-h SMPG and fasting SMPG
-
-
During the full study and maintenance periods, anytime and nocturnal confirmed hypoglycaemia were comparable
-
During the titration period (0–12 weeks), the rate of anytime and nocturnal confirmed hypoglycaemia were lower with Gla-300 vs IDeg-100
Gla-300, insulin glargine 300 U/mL; IDeg-100, insulin degludec 100 U/mL; PK/PD, pharmacokinetic/pharmacodynamic; RCT, randomized clinical trial; T2DM, type 2 diabetes; BMI, body mass index; GLP-1 RA, glucagon-like peptide-1 receptor agonist; OAD, oral antihyperglycemic drug; SMPG, self-monitored plasma glucose; SU, sulfonylureas; FPG, fasting plasma glucose; MMRM, mixed model for repeated measurements; TEAE, treatment-emergent adverse event; BL, baseline; ITT, intention-to-treat; LS, Least square; SE, standard error; W, week; CI, confidence interval; CV, coefficient of variation; SD, Standard deviation.
- Becker, R., Dahmen, R., Bergmann, K., Lehmann, A., Jax, T. and Heise, T., 2015. New Insulin Glargine 300 units {middle dot} mL-1 Provides a More Even Activity Profile and Prolonged Glycemic Control at Steady State Compared With Insulin Glargine 100 units{middle dot}mL-1. Diabetes Care, 38(4), pp.637-643.
- Heise, T., Hermanski, L., Nosek, L., Feldman, A., Rasmussen, S. and Haahr, H., 2012. Insulin degludec: four times lower pharmacodynamic variability than insulin glargine under steady‐state conditions in type 1 diabetes. Diabetes, Obesity and Metabolism, 14(9), pp.859-864.
- Ratner, R., Gough, S., Mathieu, C., Del Prato, S., Bode, B., Mersebach, H., Endahl, L. and Zinman, B., 2013. Hypoglycaemia risk with insulin degludec compared with insulin glargine in type 2 and type 1 diabetes: a pre‐planned meta‐analysis of phase 3 trials. Diabetes, Obesity and Metabolism, 15(2), pp.175-184.
- Ritzel, R., Roussel, R., Bolli, G., Vinet, L., Brulle‐Wohlhueter, C., Glezer, S. and Yki‐Järvinen, H., 2015. Patient‐level meta‐analysis of the EDITION 1, 2 and 3 studies: glycaemic control and hypoglycaemia with new insulin glargine 300 U/ml versus glargine 100 U/ml in people with type 2 diabetes. Diabetes, Obesity and Metabolism, 17(9), pp.859-867.
- Rosenstock, J., Cheng, A., Ritzel, R., Bosnyak, Z., Devisme, C., Cali, A., Sieber, J., Stella, P., Wang, X., Frías, J., Roussel, R. and Bolli, G., 2018. More Similarities Than Differences Testing Insulin Glargine 300 Units/mL Versus Insulin Degludec 100 Units/mL in Insulin-Naive Type 2 Diabetes: The Randomized Head-to-Head BRIGHT Trial. Diabetes Care, 41(10), pp.2147-2154.
- ROUSSEL, R., ROSENSTOCK, J., PETTUS, J., RITZEL, R., CHENG, A., BOSNYAK, Z., DEVISME, C., CALI, A., WANG, X. and BOLLI, G., 2018. Impact of Background Antihyperglycemic Therapy on Insulin Glargine 300 U/mL (Gla-300) vs. Insulin Degludec 100 U/mL (IDeg-100) in Insulin-Naïve People with T2DM from the BRIGHT Randomized Study. Diabetes, 67(Supplement 1), p.1029-P.
- BOLLI, G., CHENG, A., BOSNYAK, Z., CORFEC, E., CALI, A., WANG, X., FRIAS, J., ROUSSEL, R. and ROSENSTOCK, J., 2018. Lower Hypoglycemia Rates with Insulin Glargine 300 U/mL (Gla-300) vs. Insulin Degludec 100 U/mL (IDeg-100) in Insulin-Naïve Adults with T2DM on Oral Antihyperglycemic Therapy ± GLP-1RA—The BRIGHT Randomized Study. Diabetes, 67(Supplement 1), p.1032-P.
- Khunti, K., Caputo, S., Damci, T., Dzida, G., Ji, Q., Kaiser, M., Karnieli, E., Liebl, A., Ligthelm, R., Nazeri, A., Orozco-Beltran, D., Pan, C., Ross, S., Svendsen, A., Vora, J., Yale, J. and Meneghini, L., 2012. The safety and efficacy of adding once-daily insulin detemir to oral hypoglycaemic agents in patients with type 2 diabetes in a clinical practice setting in 10 countries. Diabetes, Obesity and Metabolism, 14(12), pp.1129-1136.
- Dalal, M., Kazemi, M. and Ye, F., 2016. Hypoglycemia in patients with type 2 diabetes newly initiated on basal insulin in the US in a community setting: impact on treatment discontinuation and hospitalization. Current Medical Research and Opinion, 33(2), pp.209-214.
- Mauricio, D., Meneghini, L., Seufert, J., Liao, L., Wang, H., Tong, L., Cali, A., Stella, P., Carita, P. and Khunti, K., 2017. Glycaemic control and hypoglycaemia burden in patients with type 2 diabetes initiating basal insulin in Europe and the USA. Diabetes, Obesity and Metabolism, 19(8), pp.1155-1164.


