{
event: "article_read",
name: `Odyssey outcomes - total events results`,
author: ``,
tags: `Dyslipidemia`,
publication_date: ``,
interaction_type: "content"
}
Odyssey outcomes - total events results
Odyssey Outcomes: Addition of PCSK9i to Background Statin Therapy Further Reduces MACE.
Total events study results
Study methods
- Prespecified joint semiparametric model that allows for multiple nonfatal events within a given patient
- Separate hazard functions for nonfatal and fatal events
- Association parameter quantifies the strength of the relationship between risk of nonfatal and fatal events; positive value indicates patients at high risk for nonfatal events also at high risk of death
- All analyses conducted according to intention-to-treat, including all patients and adjudicated events from randomization to the common study end date
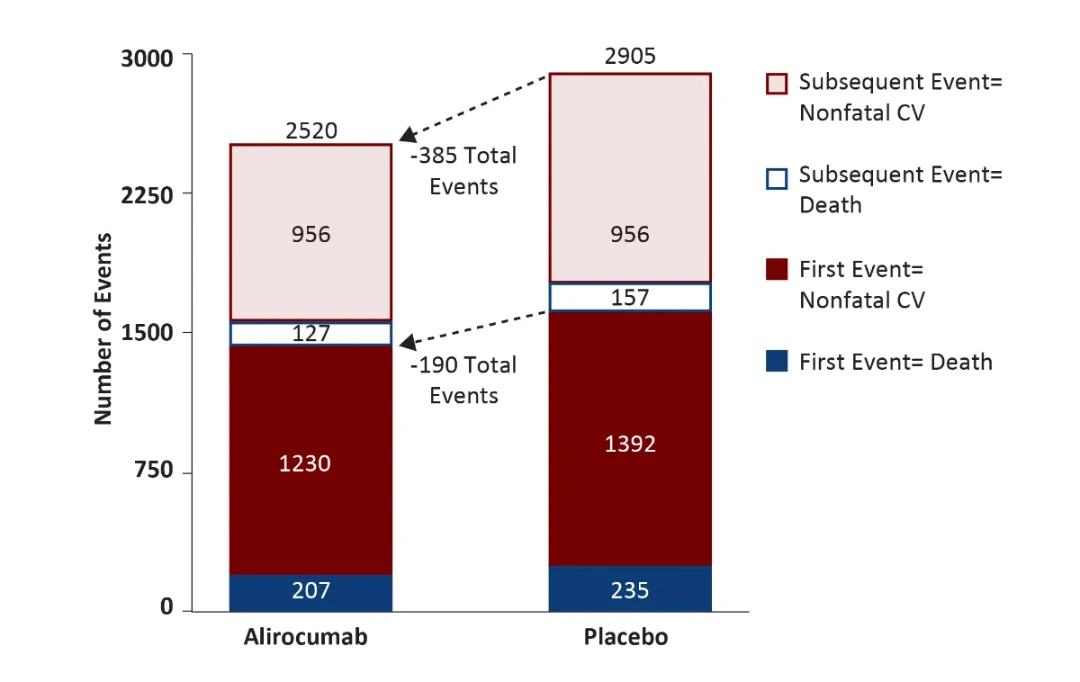
Alirocumab reduced total events
5,425 total nonfatal CV events or deaths; 77% greater than first events
Among patients with a first nonfatal CV event:
- 82%* of alirocumab patients and 85% of placebo patients were on assigned treatment; all but four alirocumab patients and three placebo patients continued treatment after first event
- 1,261 (48%) had at least one additional event
Nonfatal CV events were associated with Total Mortality
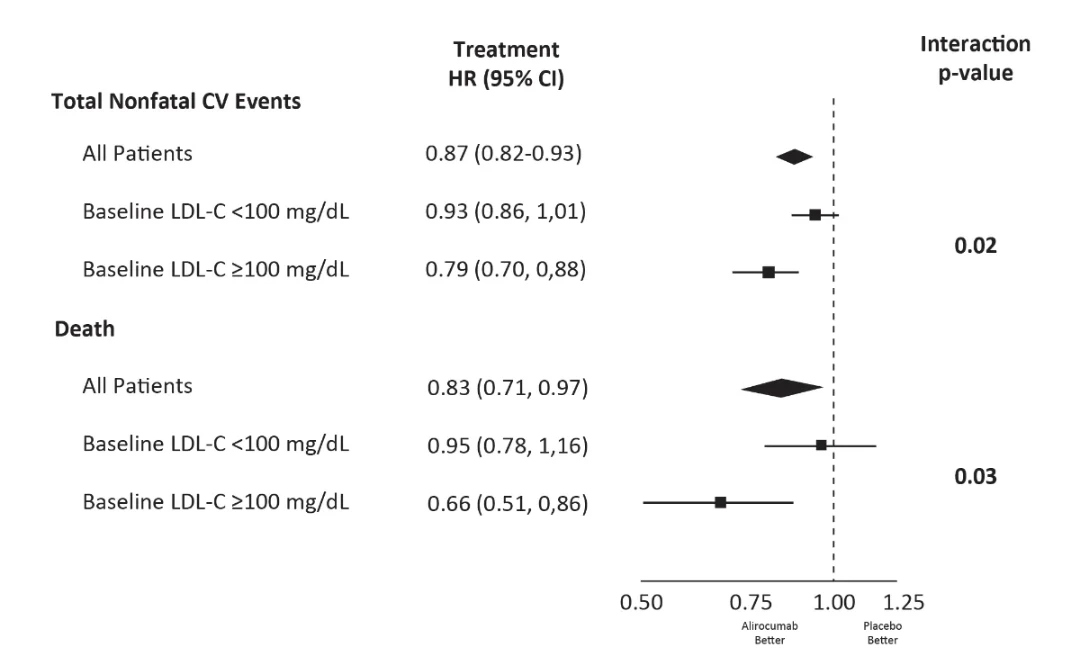
Table 4 Joint Semiparametric Models
| HR (95%CI) | p Value | |
| Death and total nonfatal cardiovascular events (n = 5,425) | ||
| Alirocumab: placebo HR for nonfatal cardiovascular events (n = 2,186 vs. n = 2,513) | 0.87 (0.82-0.93) | <0.0001 |
| Alirocumab: placebo HR for fatal events (n = 334 vs. n = 392) | 0.83 (0.71-0.97) | 0.02 |
| Association between nonfatal cardiovascular and fatal events 2.04 (95% CI: 1.78-2.29) | - | <0.0001 |
| Death and total nonfatal MI, stroke, or UA events (n = 2,999) | ||
| Alirocumab: placebo HR for nonfatal myocardial infarction, stroke, or unstable angina (n = 1,034 vs. n = 1,239) | 0.84 (0.77-0.91) | <0.0001 |
| Alirocumab: placebo HR for fatal events (n = 334 vs. n = 392) | 0.82 (0.68-0.99) | 0.04 |
| Association between nonfatal and fatal events 3.29 (95% CI: 2.86-3.72) | 0.0001 |
In relation to nonfatal events and death risk association, the post-hoc model association parameter was 1.70 (1.44, 1.96), p<0.0001
Higher baseline LDL-C associated with greater reduction of total events with Alirocumab
- 255 fewer total events with alirocumab among 5,629 patients with LDL-C >100 mg/dl at baseline
- 130 fewer total events with alirocumab among 13,295 patients with LDL-C
Total events study conclusion
- In the previously reported primary analysis of the study data, the observed 15% relative risk reduction (RRR) in allcause death with alirocumab was considered nominally significant due to the prespecified testing sequence of secondary endpoints
- The joint models showed an associated reduction in non-fatal CV events and was a predictor of non-fatal CV mortality by alirocumab
- The authors in the manuscript conclude that this complementary modelling strategy therefore supports the observation that alirocumab reduced all-cause death in the trial
- They further discuss that reduction in mortality risk may be viewed as a preferred metric to summarise the observed effect of treatment with alirocumab on mortality
- Szarek M, et al. JACC. 2019;73(4):387-96
Related articles
MAT-BH-2100555/v2/Jun 2023



.png/jcr:content/lab%20thumb%20(1).png)
.webp/jcr:content/science%20thumb%20(2).webp)