VTE prophylaxis in medically ill patients
The Medenox Study: Prophylaxis in medical patients with Enoxaparin
Objective
Evaluating the efficacy and safety of enoxaparin for the prophylaxis of venous thromboembolism (VTE) in acute medical patients.¹
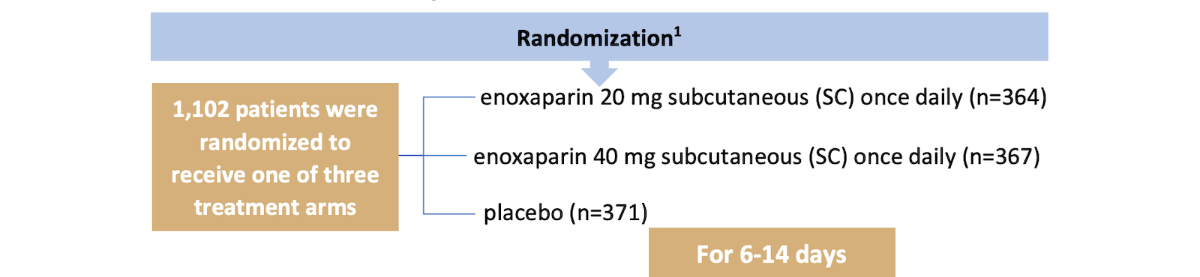
Study design: Screening¹
Phase III double-blind, placebo-controlled, multi-center trial
Patients were primarily hospitalized for acute heart failure (New York Heart Association [NYHA] Class III/IV) or acute respiratory failure. Subjects with at least one predefined VTE risk factor plus 1 of the following medical conditions were also enrolled:
- Acute Rheumatic Disorders
- Acute Infection (without septic shock)
- Active episodes of inflammatory bowel
Risk factors included
The primary endpoint:¹ The development of VTE (deep vein thrombosis [DVT], pulmonary embolism [PE], or both) between days 1 and 14.
The secondary endpoint: The occurrence of VTE between days 1 and 110.
Baseline characteristics¹
- Baseline characteristics were similar between the enoxaparin and placebo groups.
- The mean number of risk factors per patient was 2.1 in the placebo group, 2.0 in the enoxaparin 20 mg group, and 2.1 in the enoxaparin 40 mg group.
- Median treatment duration was 7 days and did not differ significantly between the groups
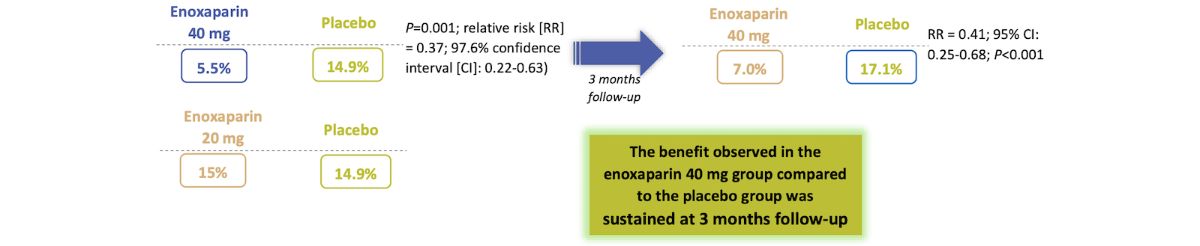
Results: Efficacy¹
The intent-to-treat analysis demonstrated a statistically significant reduction in the rate of VTE between days 1-14 in the enoxaparin 40 mg group compared to placebo, while enoxaparin 20 mg showed comparable results to placebo.
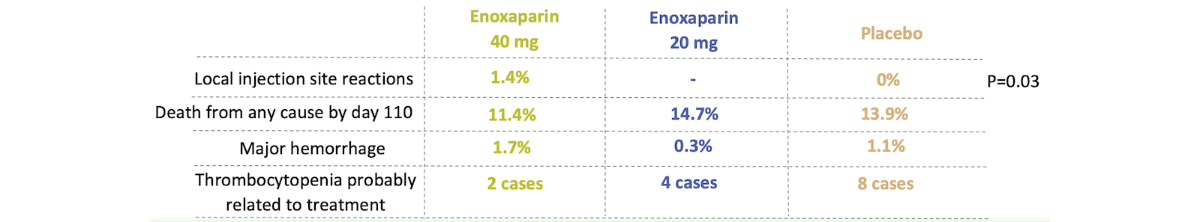
Results: Safety¹
No significant difference in the rate of adverse events (AEs) was noted between placebo and either enoxaparin group with the exception of local injection site reactions.
The investigators concluded that enoxaparin 40 mg SC daily was effective and safe in reducing the risk of VTE in acutely-ill hospitalized medical patients.1
Post-hoc analysis of Medenox study*²
The relative risk reductions (RRR) for VTE in the enoxaparin 40 mg group compared to placebo in the disease subgroups and in the pre-defined risk factor subgroups.
|
Disease State |
Medenox study (n) |
Placebo |
Enoxaparin 40 mg |
Relative Risk |
P value |
|---|---|---|---|---|---|
| Heart failure | 290 | 14.6%(14/96) | 4.0% (4/99) | 0.29 (0.10-0.84) | 0.02 |
| Class III | 217 | 12.3%(9/73) | 5.1% (4/78) | 0.42 (0.13-1.29) | 0.2 |
| Class IV | 73 | 21.7%(5/23) | 0% (0/21) | - | 0.05 |
| Respiratory disease | 457 | 13.1%(20/153) | 3.3% (5/153) | 0.25 (0.10-0.65) | 0.003 |
| Infectious disease | 463 | 15.5%(24/155) | 6.3%(10/159) | 0.41 (0.20-0.82) | 0.01 |
| Infectious and respiratory disease | 239 | 16.5%(13/79) | 4.6% (4/88) | 0.28 (0.09-0.81) | 0.02 |
| Rheumatic disease | 78 | 20.7%(6/29) | 10.0%(2/20) | 0.48 (0.11-2.16) | 0.4 |
| Inflammatory Bowel disease | 5 | 0% (0/1) | 0% (0/3) | - | - |
| Pre-defined risk factor | Relative Risk (confidence interval) | P value |
|---|---|---|
| Chronic heart failure | 0.26 (0.08-0.92) | 0.04 |
| Chronic respiratory disease | 0.26 (0.10-0.68) | 0.005 |
| Age | 0.22 (0.09-0.51) | 0.0001 |
| Immobility | 0.44 (0.22-0.88) | 0.02 |
| Previous VTE | 0.49 (0.15-1.68) | 0.4 |
| Cancer | 0.50 (0.14-1.72) | 0.4 |
| Obesity | 0.49 (0.18-1.36) | 0.3 |
A subgroup analysis of the Medenox study was conducted to determine the patient characteristics of medical patient populations that benefit from prophylaxis with enoxaparin 40 mg compared to placebo in the prevention of VTE. RR was calculated from the 40 mg group data for each of the major disease groups and for eight risk factors that had been recorded in the Medenox study²
The authors concluded that enoxaparin was significantly superior to placebo in the prevention of VTE in patients hospitalized with heart failure (NYHA class III or IV), acute respiratory failure, and acute infectious diseases. Overall, the benefits of enoxaparin extended to a range of acutely ill medical patients.²
VTE, Venous thromboembolism
- Samama, M., Cohen, a., Darmon, J., Desjardins, L., Eldor, a., Janbon, C., Leizorovicz, a., Nguyen, H., Olsson, C., Turpie, a. And Weisslinger, N., 1999. A Comparison of Enoxaparin With Placebo for the Prevention of Venous Thromboembolism in Acutely Ill Medical Patients. The New England Journal of Medicine, 341(11), Pp.793-800.
- Alikhan, R., Cohen, A., Combe, S., Samama, M., Desjardins, L., Eldor, A., Janbon, C., Leizorovicz, A., Olsson, C. and Turpie, A., 2003. Prevention of venous thromboembolism in medical patients with enoxaparin: a subgroup analysis of the Medenox study. Blood Coagulation and Fibrinolysis, 14(4), pp.341-346.