Dupilumab Efficacy Across Baseline Eosinophil Counts in COPD
Dupilumab Efficacy in COPD Patients with Type 2 Inflammation: Post Hoc Analysis Stratified by Baseline
Blood Eosinophil Counts (≥150 vs ≥300 cells/μL) from Pooled BOREAS and NOTUS Data
Dupilumab Efficacy in Patients With
COPD With Type 2 Inflammation
Across Baseline Eosinophil Counts
Stephanie A. Christenson, Frank C. Sciurba, Claus F. Vogelmeier, Marc Miravitlles, John R. Hurst, Fuqiang Wen, Fernando J. Martinez, Paramita Saha-Chaudhuri, Mena Soliman, Jigna Heble
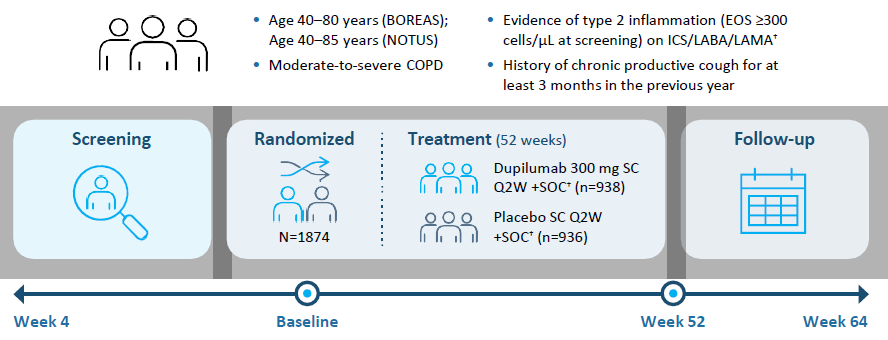
Study Design* |
Background: Due to variability in BEC in COPD patients and uncertainty around whether current or historical BEC ≥300 cells/ μL is more predictive of treatment response, we evaluated whether dupilumab remained effective in patients whose BEC declined between screening and randomization
Study Objective: This post hoc analysis of pooled BOREAS and NOTUS data evaluated dupilumab efficacy in COPD patients with type 2 inflammation, stratified by baseline blood EOS counts (≥150 or ≥300 cells/μL)
Endpoints |
*All patients had blood EOS counts of ≥300 cells/μL at screening. †Triple therapy (ICS+LABA+LAMA) for 3 months before randomization with a stable dose of medication for ≥1 month before Visit 1; dual therapy (LABA+LAMA) allowed if ICS contraindicated.
BD, bronchodilator; BEC, blood eosinophil count; COPD, chronic obstructive pulmonary disease; EOS, eosinophil; FEV1, forced expiratory volume in 1 second; ICS, inhaled corticosteroid; ITT, intent-to-treat; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; Q2W, every 2 weeks; SC, subcutaneous; SGRQ, St. George’s Respiratory Questionnaire; SOC, standard of care.
Christenson SA, et al. Poster presented at: American Thoracic Society International Congress; May 16-21, 2025; San Francisco, CA, USA.
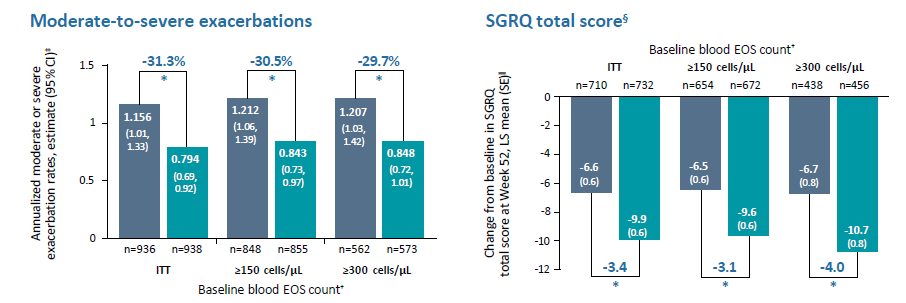
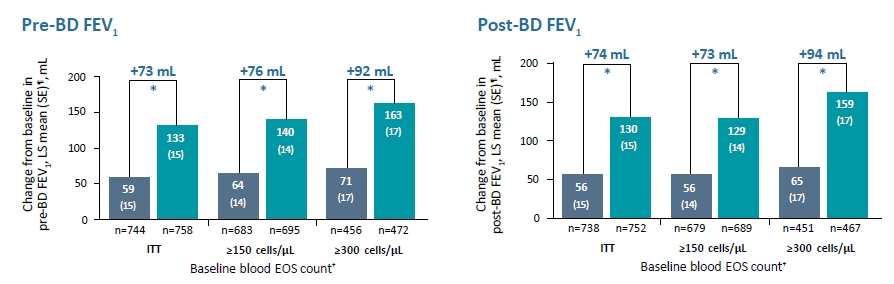
Results |
*P<0.001 vs placebo
Conclusion |
In patients with COPD and type 2
inflammation, including the ITT population
and those with baseline EOS counts ≥150
and ≥300 cells/μL, dupilumab:
†All patients had blood EOS counts of ≥300 cells/μL at screening. ‡Derived using negative binomial model with the total number of events occurring during the 52-week treatment period as the response variable, and treatment group, region (pooled country), ICS dose at baseline (high dose ICS [yes/no]), smoking status at screening (current smokers or not), baseline disease severity (as % predicted post- bronchodilator FEV1), and number of moderate or severe COPD exacerbation events within one year prior to the study (≤2, 3, or ≥4) as covariates. Log-transformed observation duration will be used as offset variable. §SGRQ total scores range from 0 to 100, with lower scores indicating a better quality of life. ǁDerived from an MMRM model with the change from baseline in SGRQ total score up to Week 52 as a response variable, and treatment group, study (if pooled), region (pooled country), ICS dose, smoking status at screening, treatment-by-visit interaction, baseline SGRQ total score, and SGRQ baseline-by-visit interaction as covariates. ¶Derived from an MMRM model with the change from baseline in pre-/post-bronchodilator FEV1 up to Week 52 as a response variable, and treatment group, study, age, sex, height, region (pooled country), ICS dose, smoking status at screening, visit, treatment-by-visit interaction, baseline pre-/post-bronchodilator FEV1, and FEV1 baseline-by-visit interaction as covariates.
BD, brochodilator; CI, confidence interval; COPD, chronic obstructive pulmonary disease; EOS, eosinophil; FEV1, forced expiratory volume in 1 second; ITT, intent-to-treat; LS, least square; MMRM, mixed model of repeated measures; SE, standard error; SGRQ, St. George’s Respiratory Questionnaire.
Christenson SA, et al. Poster presented at: American Thoracic Society International Congress; May 16-21, 2025; San Francisco, CA, USA.