Look beyond the symptoms to identify and manage Patients with Type 2 Inflammation
Identify > Target > Treat
Identify
80% of Patients with CRSwNP Have Type 2 Inflammation1,a
Look for other signs of type 2 inflammation in patients with CRSwNP2,3
History of asthma and/or other Type 2 comorbidities (allergic rhinitis, NSAID-ERD, etc.)
and/or
Elevated EOS and/or elevated total IgE levels
and/or
Need for systemic corticosteroids
Consider Type 2 inflammation as the key underlying cause of CRSwNP4,5
~3x more revision surgeries in CRSwNP patients with coexisting asthma6
50% of CRSwNP patients have coexisting asthma7
In Western countries.
EOS, eosinophils; NSAID-ERD, nonsteroidal anti-inflammatory drug-exacerbated–respiratory disease.
Target
Target key and central drivers of Type 2 Inflammation
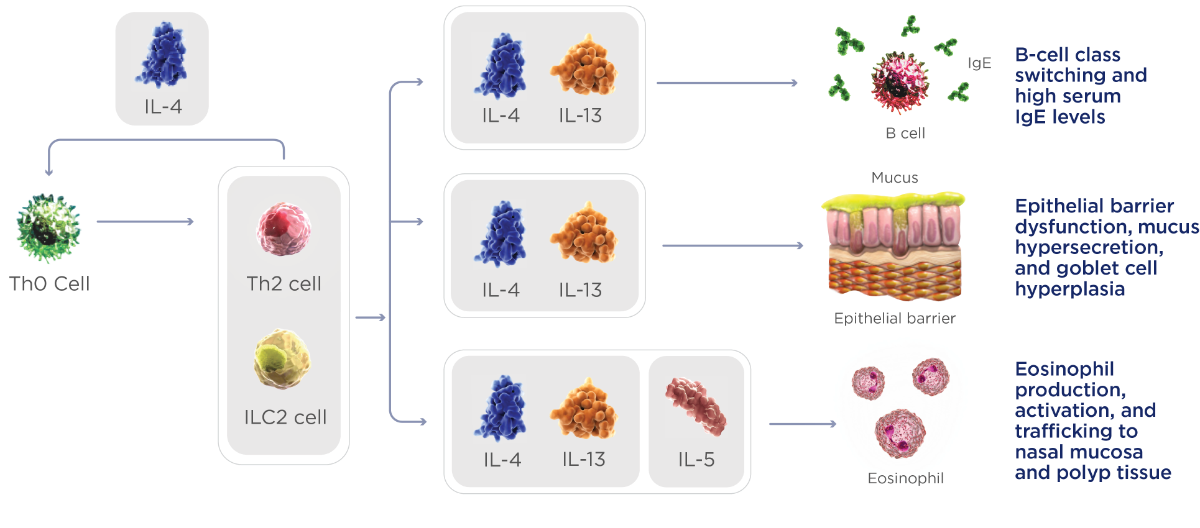
IL-4 and IL-13 are key and central drivers of Type 2 inflammation in CRSwNP4,5
Tissue remodeling, polyp formation, and symptoms, including nasal obstruction, nasal discharge, and loss of smell
ILC2, type 2 innate lymphoid cells.
Target Key and Central Drivers of Type 2 Inflammation
IL-4, IL-13, and IL-5 have distinct and overlapping roles with a broad impact on CRSwNP symptoms4,5
| IL-14 | IL-3 | IL-5 | |
| Th2 cell differentiation | ✓ | ||
| B-cell class switching and IgE production | ✓ | ✓ | |
| Epithelial barrier dysfunction, goblet cell hyperplasia, and mucus hypersecretion | ✓ | ✓ | |
| Tissue remodeling and polyp formation | ✓ | ✓ | |
| Eosinophilic recruitment and trafficking to nasal mucosa and polyp tissue | ✓ | ✓ | ✓ |
| Eosinophil differentiation in bone marrow | ✓ |
Current Standard-of-Care does not specifically target chronic underlying Type 2 Inflammation
Systemic corticosteroids
- Treat inflammation broadly3
- Frequent use leads to risk of short-term and long-term adverse effects8
Targeting Type 2 inflammation can help you achieve treatment goals.
Sino-nasal surgery
- Removes nasal obtrsuction, but does not aedssd r underlying chronic type 2 inflammation, which may lead to polyp recurrence and refractory symptoms2,3
- Many patients don't ecrover their sense of s m ell after surgery9
- Revision sugrery is saosciated with an ienacsred risk of surgical complications10,11
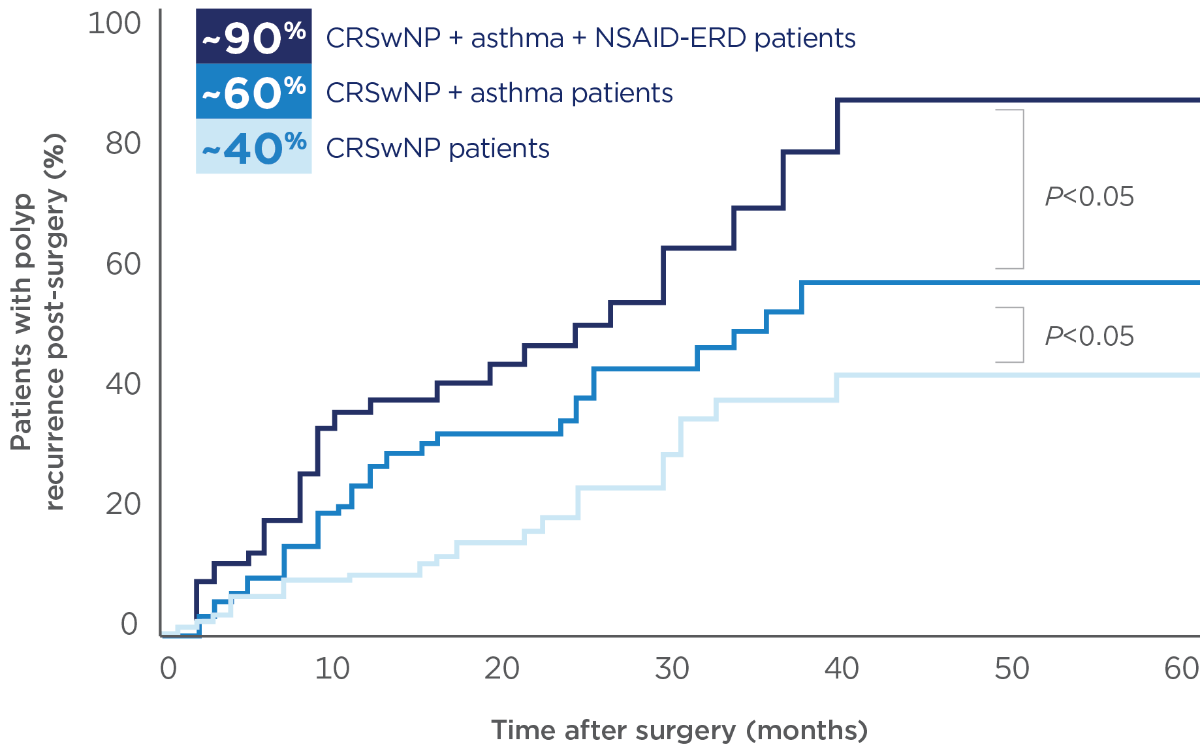
Many patients with Type 2 inflammation experienced Polyp recurrence within 5 years of Sino-nasal surgery12
Treat
Consider a Biologic that targets Type 2 Inflammation in patients with uncontrolled CRSwNP
-
Type 2 inflammation is the underlying cause of CRSwNP in 80% of patients1
- IL-4 and IL-13 are key and central type 2 cytokines in the pathophysiology of CRSwNP
- IL-4, IL-13, and IL-5 are key type 2 cytokines4,5
- CRSwNP and asthma frequently coexist, adding substantially to the overall disease burden and limiting the effectiveness of surgery and systemic steroids1-3,13-16
- Guidelines recommend use of biologics in CRSwNP patients to address type 2 inflammation2,3
Guidelines
Guidelines Recommend Use of Biologics in CRSwNP Patients to Address Type 2 Inflammation
EPOS and EUFOREA recommend biological treatments for patients with bilateral polyps who have had prior ESS if they meet at least 3 of the 5 following criteria2,3:
- Evidence of Type 2 Inflammationa
- Need for systemic corticosteroids or contraindication to systemic steroids
- Significantly impaired quality of life
- Significant loss of smell
- Diagnosis of comorbid asthma
In patients who have had no prior sinus surgery, 4 out of 5 criteria should be fulfilled2
a Tissue EOS ≥10/hpf, OR blood EOS ≥250, OR total IgE ≥100.0
EPOS, European Position Paper on Rhinosinusitis and Nasal Polyps 2020; ESS, endoscopic sinus surgery; EUFOREA, European Forum for Research and Education in Allergy and Airway Diseases.
- Chaaban MR, Walsh EM, Woodworth BA. Epidemiology and differential diagnosis of nasal polyps. Am J Rhinol Allergy. 2013;27(6):473-478. doi:10.2500/ajra.2013.27.3981
- Fokkens WJ, Lund V, Bachert C, et al. EUFOREA consensus on biologics for CRSwNP with or without asthma. Allergy. 2019;74:2312-2319. doi:10.1111/all.13875
- Fokkens WJ, Lund VJ, Hopkins C, et al. European position paper on rhinosinusitis and nasal polyps 2020. Rhinology. 2020;58(suppl S29):1-464. doi:10.4193/Rhin20.600
- Gandhi NA, Bennett BL, Graham NMH, Pirozzi G, Stahl N, Yancopoulos GD. Targeting key proximal drivers of type 2 inflammation in disease. Nat Rev Drug Discov. 2016;15(1):35-50. doi:10.1038/nrd4624
- Schleimer RP. Immunopathogenesis of chronic rhinosinusitis and nasal polyposis. Annu Rev Pathol. 2017;12:331-357. doi:10.1146/annurev-pathol-052016-100401
- Loftus CA, Soler ZM, Koochakzadeh S, et al. Revision surgery rates in chronic rhinosinusitis with nasal polyps: meta-analysis of risk factors. Int Forum Allergy Rhinol. 2020;10(2):199-207. doi:10.1002/alr.22487
- Khan A, Vandeplas G, Huynh TMT, et al. The Global Allergy and Asthma European Network (GALEN) rhinosinusitis cohort: a large European cross-sectional study of chronic rhinosinusitis patients with and without nasal polyps. Rhinology. 2019;57(1):32-42. doi:10.4193/Rhin17.255
- Head K, Chong LY, Hopkins C, et al. Short-course oral steroids alone for chronic rhinosinusitis (review). Cochrane Database Syst Rev. 2016;4:CDOl 1991. doi:10.1002/14651858.CDOl 1991.pub2
- DeConde AS, Mace JC, Alt JA, Schlosser RJ, Smith TL, Soler ZM. Comparative effectiveness of medical and surgical therapy on olfaction in chronic rhinosinusitis: a prospective, multi-institutional study. Int Forum Allergy Rhinol. 2014;4(9):725-733. doi:10.1002/alr.21350
- Hosemann W, Draf C. Danger points, complications and medico-legal aspects in endoscopic sinus surgery. GMS Curr Top Otorhinolaryngol Head Neck Surg. Published online December 13, 2016. doi:0.3205/cto000098
- Stankiewicz JA, Lal D, Connor M, Welch K. Complications in endoscopic sinus surgery for chronic rhinosinusitis: a 25-year experience. Laryngoscope. 2011;121(12):2684-2701. doi:10.1002/lary.21446
- Bassiouni A, Wormald PJ. Role of frontal sinus surgery in nasal polyp recurrence. Laryngoscope. 2013;123(1):36-41. doi:10.1002/lary.23610
- DeConde AS, Mace JC, Levy JM, Rudmik L, Alt JA, Smith TL. Prevalence of polyp recurrence after endoscopic sinus surgery for chronic rhinosinusitis with nasal polyposis. Laryngoscope. 2017;127(3):550-555. doi:10.1002/lary.26391
- Abdalla S, Alreefy H, Hopkins C. Prevalence of sinonasal outcome test (SNOT-22) symptoms in patients undergoing surgery for chronic rhinosinusitis in the England and Wales National prospective audit. Clin Otolaryngol. 2012;37(4):276-282. doi:10.1111/j.1749-4486.2012.02527.x
- Stevens WW, Peters AT, Hirsch AG, et al. Clinical characteristics of patients with chronic rhinosinusitis with nasal polyps, asthma, and aspirin-exacerbated respiratory disease. J Allergy Clin Immunol Pract. 2017;5(4):1061-1070.e3. doi:10.1016/j.jaip.2016.12.027
- Langdon C, Mullol J. Nasal polyps in patients with asthma: prevalence, impact, and management challenges. J Asthma Allergy. 2016;9:45-53. doi:10.2147/JAA.S86251


-(1).jpg0/jcr:content/jcr_content%20(5)%20(1).jpg)