FLUZONE® Trivalent
For patients 6 months of age and older

A trivalent influenza vaccine option for children and adults*
FLUZONE® Influenza Virus Vaccine Trivalent Types A and B (Split Virion) is indicated for active immunization against influenza caused by the specific strains of influenza virus contained in the vaccine in adults and children 6 months of age and older.1
About FLUZONE® Trivalent
FLUZONE® Trivalent is a split virion inactivated influenza virus vaccine consisting of a sterile suspension containing three strains of influenza viruses propagated in embryonated chicken eggs.*
Dosage forms and ingredients*

FLUZONE® Trivalent is supplied in single-dose prefilled syringes and multi-dose vials.
The vaccine is available in packages of:
- 1 x 5 mL (multi-dose) vial
- 10 x 0.5 mL (single dose) syringes without attached needle

This vaccine complies with the World Health Organization (WHO) recommendation (Northern Hemisphere) for the 2025-2026 season.
Each 0.5 mL dose is formulated to contain 15 μg of hemagglutinin (HA) of each strain listed below.
A (H3N2)
A (H1N1)
B/strain
Non-medicinal ingredients:
0.5 mL dose
Phosphate Buffered Saline [sodium chloride 6.51 g/L;
sodium phosphate (dibasic anhydrous) 3.83 g/L;
sodium phosphate (monobasic anhydrous) 0.410 g/L]
Water for injection quantity sufficient up to 0.5 mL
Triton® X-100 NMT 250 mcg
0.01% w/v thimerosal is present in multi-dose presentation only
* Clinical significance has not been established.
Dosage and administration
Patients 6 months through 35 months
1 or 2 dosesa, 4 weeks apartb 0.25 mLc or 0.5 mLd
Patients 3 through 8 years
1 or 2 dosesa 0.5 mL
Patients 9 years and up
1 dose 0.5 mL
NOTE: Fractional doses (doses of less volume than indicated for each age group above) should not be given.
a Previously unvaccinated children 6 months to <9 years of age require 2 doses of seasonal influenza vaccine with an interval of 4 weeks. Eligible children <9 years of age who have properly received one or more doses of seasonal influenza vaccine in the past are recommended to receive one dose per season thereafter.
b If a child’s second dose is missed, it can be given at any time.
c In clinical studies, children 6 months through 35 months received a 0.25 mL dose.
d NACI recommends that children 6 months through 35 months should be given a full 0.5 mL dose of influenza vaccine.
Injection steps
- Do not administer by intravascular injection. Administer the vaccine intramuscularly.
- FLUZONE® Trivalent should not be administered into the buttocks. The preferred site is into the deltoid muscle in adults and children >1 year of age. The preferred site for infants and children <1 year of age is the anterolateral aspect of the mid-thigh.
- Shake vial or prefilled syringe well before administering each dose.
- Aseptic technique must be used. Use a separate sterile needle and syringe (or sterile prefilled syringe) for each individual patient and for each entry into a multi-dose vial. Needles should not be recapped and should be disposed of according to biohazard waste guidelines.
- Give the patient a permanent personal immunization record. In addition, it is essential that the physician or nurse record the immunization history in the permanent medical record of each patient. This permanent office record should contain the name of the vaccine, date given, dose, manufacturer and lot number.
Safety profile
The strain composition of the influenza virus vaccines is subject to annual changes. Respective clinical studies, including at least 50 adults 18–60 years of age and at least 50 older adults aged 60 years or older, are conducted to assess the safety and immunogenicity of FLUZONE®. For the purpose of the cumulative analysis, five years of annual clinical safety data are presented below. In this data set, a total of 601 vaccinees received an intramuscular injection of FLUZONE®.
Percentage of Solicited Injection-Site Reactions and Systemic Adverse Events in Children and Adults After Vaccination with FLUZONE® Trivalent
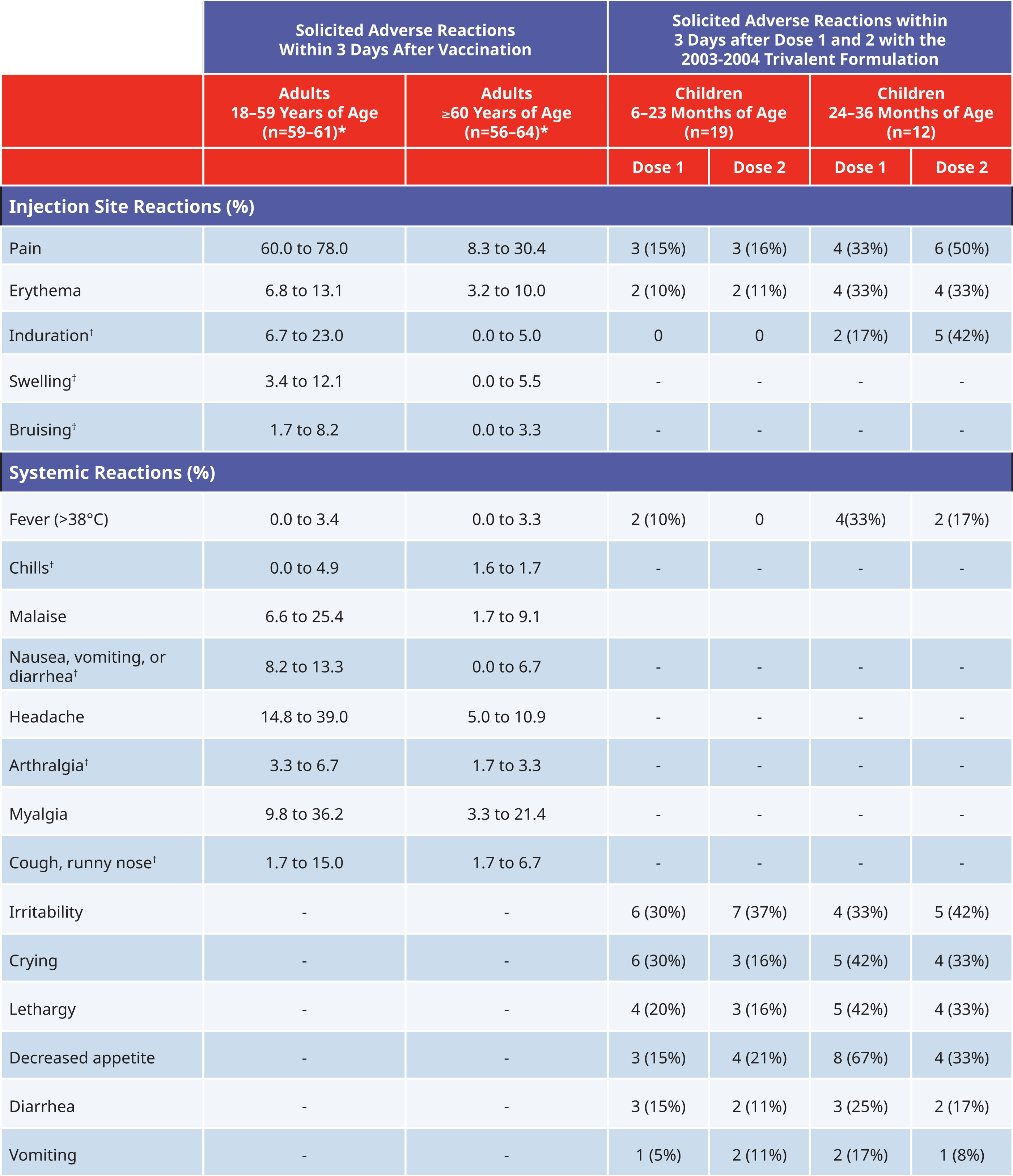
* Safety population analyzed per study
† Specific adverse reactions were not reported or solicited in all studies
Safety information
CONTRAINDICATIONS:
Should not be administered to anyone with with a history of severe allergic reaction to egg protein or any component of the vaccine or after previous administration of the vaccine or a vaccine containing the same components or constituents.
CLINICAL USE:
Pediatrics (6 months – 18 years of age): Based on the data submitted and reviewed by Health Canada, the safety and efficacy of FLUZONE® in pediatric patients have been established. Therefore, Health Canada has authorized an indication for pediatric use.
Safety and efficacy of FLUZONE® administration in children less than 6 months of age have not been established.
Geriatrics: Evidence from clinical studies and experience suggests that use in the geriatric population is associated with differences in safety or effectiveness.
RELEVANT WARNINGS & PRECAUTIONS:
- Do not administer by intravascular injection. Do not administer into the buttocks.
- As with any vaccine, immunization with FLUZONE® Trivalent may not protect 100% of individuals.
- Vaccination should be postponed in case of a moderate or severe acute disease with or without fever: however in case of mild disease with low-grade fever, vaccine could be administered
- FLUZONE® Trivalent should not be administered to persons with bleeding disorders such as hemophilia or thrombocytopenia, or in persons on anticoagulant therapy, unless the potential benefits outweigh the risk of administration. If the decision is made to administer any product by intramuscular injection to such persons, it should be given with caution, with steps taken to avoid hematoma formation risk following injection.
- Immunocompromised persons may not achieve the expected immune response.
- Immunization should be delayed in a patient with an active neurologic disorder but should be considered when the disease process has been stabilized.
- Guillain-Barré syndrome (GBS) has been reported after influenza vaccination. However, it is not known whether influenza vaccination specifically might increase the risk for recurrence of GBS. Therefore, NACI and the US Advisory Committee on Immunization Practices (ACIP) state it is prudent to avoid vaccinating persons who are not at high risk for severe influenza complications and who are known to have experienced GBS within 6 weeks after a previous influenza vaccination.
- Syncope can occur following, or even before, any vaccination as a psychogenic response to the needle injection. Procedures should be in place to prevent falling and injury and to manage syncope.
-
Animal reproductive studies have not been conducted with FLUZONE®. It is also not known whether FLUZONE® can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Data on the use of this vaccine in pregnant women are limited. FLUZONE® should be given to pregnant women only if clearly needed and following an assessment of the risks and benefits. However, there is no evidence to suggest a risk to the fetus or the pregnancy from maternal immunization with FLUZONE®. NACI states that influenza vaccination is recommended for pregnant women
- There are no available data on the presence of FLUZONE® Trivalent in human milk, effects on milk production or the effects on the breastfed infant. No conclusions can be drawn regarding whether or not FLUZONE® Trivalent is safe for use during breastfeeding. FLUZONE® Trivalent should be used during breastfeeding only if the potential benefits to the mother outweigh the potential risks including those to the breastfed child. NACI states that influenza vaccination is considered safe for breastfeeding women.
FOR MORE INFORMATION:
Consult the Product Monograph for important information relating to adverse reactions, drug interactions, and dosing information which have not been discussed in this piece. The product monograph is also available through our medical department.
Call us at 1-800-265-7927.
- Product Monograph: FLUZONE® High-Dose Trivalent. Sanofi Pasteur. February 13, 2025.
- An Advisory Committee Statement (ACS)/National Advisory Committee on Immunization (NACI): Canadian Immunization Guide Chapter on Influenza and Statement on Seasonal Influenza Vaccine for 2024–2025. May 2024.
MAT-CA-2500261-v.2 09/2025


.jpg/jcr:content/image%20(1).jpg)
.jpg/jcr:content/image%20(2).jpg)
.jpg/jcr:content/image%20(3).jpg)