Airway remodeling in asthma: Why targeting IL-4 and IL-13 matters1,9
Airway remodeling drives progressive and potentially irreversible lung function decline in asthma.2 Understanding the central role of IL-4 and IL-13 in this process is essential to guiding effective treatment strategies.1,10
Listen to Prof. Alberto Papi about Airway remodeling
Understanding airway remodeling and hyperresponsiveness1,3-5
Asthma involves chronic airway inflammation, structural changes in the lung airway lining, and exaggerated airway constriction in response to stimuli.3,4 These processes — airway remodeling and airway hyperresponsiveness (AHR) — are the key underlying mechanisms of lung function decline.1,5
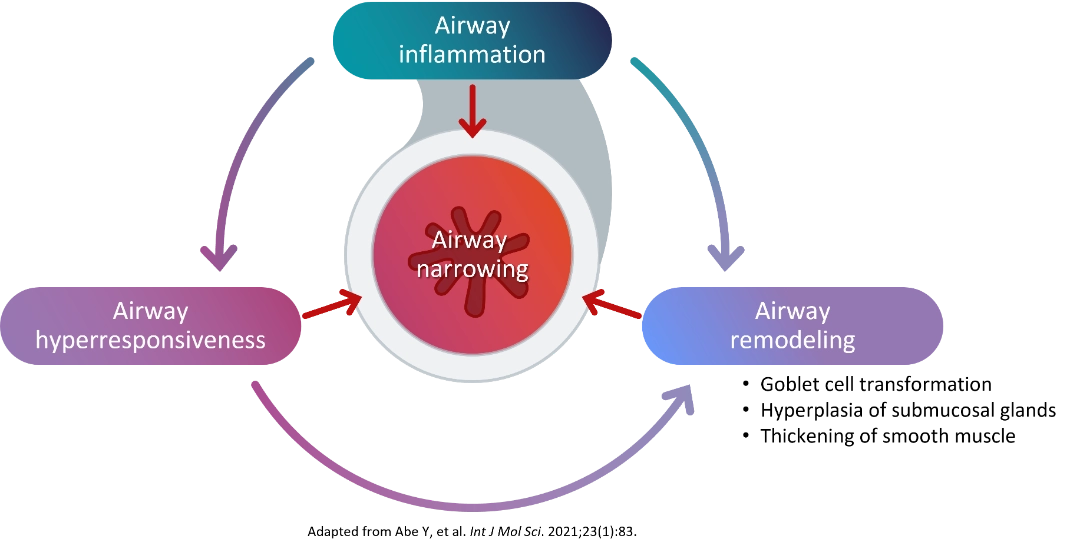
Type 2 inflammation, Hyperresponsiveness, and Remodeling contribute to Airway narrowing6 |
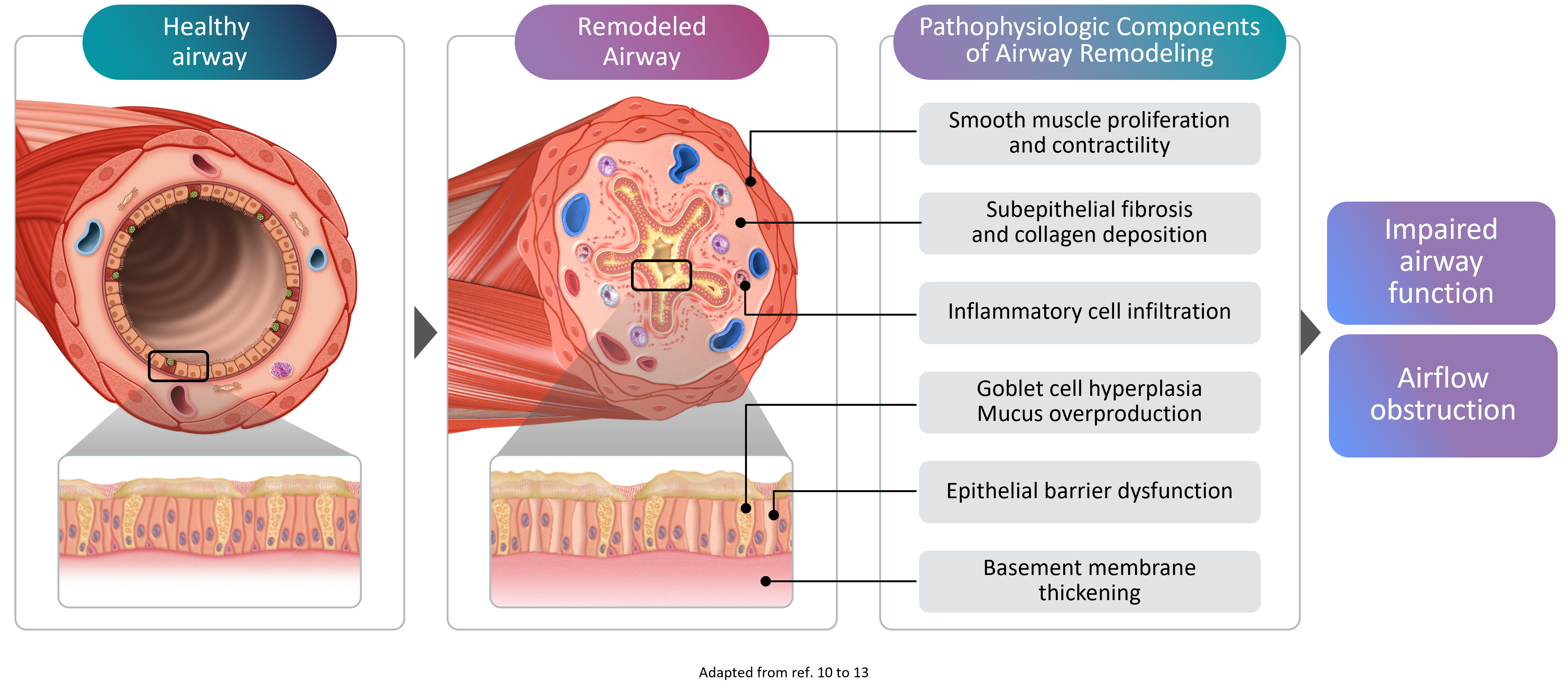
Airway remodeling refers to the progressive structural changes in the airways that occur as a result of chronic inflammation. These changes include:
- Thickening of the reticular basement membrane - the layer beneath the airway lining becomes abnormally thick
- Subepithelial fibrosis - excess scar tissue forms beneath the airway surface
- Hypertrophic airway smooth muscle (ASM) cells - the muscle surrounding the airways increases in size, narrowing the airway lumen
- Goblet cell hyperplasia - mucus-producing cells multiply, leading to excessive mucus production4
Remodeling in asthma comprises structural changes to the airways10-13
AHR is the exaggerated tendency of the airways to constrict in response to stimuli. The mechanisms underlying AHR include increased contractility of ASM, persistent airway inflammation, and structural changes from airway remodeling.3,5
Clinically, AHR contributes to:
- Accelerated lung function decline over time
- Recurrent wheezing and persistent symptoms
- Increased frequency and severity of exacerbations5
Key clinical insight |
AHR is associated with increased risk of lung function decline, persistence of symptoms such as wheezing, and occurrence of future exacerbations. Treatment strategies focused on normalization of AHR can reduce exacerbation risk and prevent further airway inflammation and remodeling.5 |
The role of type 2 inflammation10
Majority of patients with asthma have type 2 inflammation as the main causative pathophysiologic process.10 This type of asthma is driven by type 2 cytokines (primarily IL-4, IL-13, and IL-5) with additional contributions from alarmins like thymic stromal lymphopoietin (TSLP), an epithelial-derived regulator of type 2 inflammation.10
How type 2 cytokines drive asthma pathology?1-2,10-11
The cumulative action of IL-4 and IL-13 causes edema, mucus hypersecretion, AHR, and airway remodeling - ultimately leading to decreased lung function.10
Did you know? |
Eosinophils are NOT the primary drivers of airway remodeling. IL-4 and IL-13 are the cytokines mainly responsible for the structural airway changes that lead to lung function decline. Addressing these cytokines is essential to halt lung damage.1,2 |
What is the role of each type 2 cytokine in the process of airway remodeling?
IL-4 |
IL-13 |
IL-5 |
| Differentiation of T-cell and B-cell | ||
|
|
Differentiation of eosinophil in bone marrow | |
| Migration and extravasation of eosinophils into the lung airways | ||
| IgE production and class switching; degranulation of mast cell and basophil |
||
| Goblet cell maturation; mucus secretion |
||
| Differentiation of bronchial epithelial cells | ||
| Increased contractility of airway smooth muscle cells, and bronchial hyperresponsiveness | ||
Note: The thymic stromal lymphopoietin (TSLP), an epithelium-derived cytokine, contributes to airway remodeling in lungs by facilitating differentiation of T helper 2 (Th2) cells, and influences dendritic cell function.
IL-4 and IL-13: The critical therapeutic targets1,8,9
While eosinophil count and IL-5 are important markers of asthma exacerbations, IL-4 and IL-13 are the primary drivers of the structural airway changes seen in remodeling.2 Therapeutic approaches targeting IL-4 and IL-13 can address both the inflammatory cascade and the underlying remodeling processes that contribute to lung function decline.2
Research evidence supports the direct role of IL-4 and IL-13 in airway inflammation, AHR, and airway remodeling — making them critical therapeutic targets.1,8,9
Comparison of therapeutic targets
Target |
Mechanism |
Impact on Airway Remodeling |
| IL-4 & IL-131,2,9 | Drive airway inflammation, eosinophil migration and adhesion, mucus hypersecretion, and AHR. | Direct — address the root cause of remodeling and lung function decline. |
| IL-51,2 | Mediates eosinophil differentiation in bone marrow and activation in airways. | Secondary role — influences remodeling through eosinophil-mediated inflammatory processes. |
Blocking IL-4 and IL-13 can also suppress eosinophilic inflammation indirectly — reducing airway tissue eosinophilia.1
Key message |
Blocking IL-4 and IL-13 can reduce airway tissue eosinophilia, decrease immune infiltrates associated with type 2 inflammation, and extend therapeutic benefits beyond eosinophil count reduction alone.1,2 |
Clinical implications: The importance of early intervention7
Airway remodeling is a progressive process. Early detection and intervention targeting the underlying inflammatory drivers can reduce acute asthma attacks and minimize irreversible lung function decline.7
Identifying the type of inflammation present in each patient is key to effective disease management.
The following type 2 inflammation biomarkers should be assessed:7
- Blood eosinophil count — elevated levels indicate eosinophilic airway inflammation
- Fractional exhaled nitric oxide (FeNO) — a marker of IL-13-driven airway inflammation
- Serum IgE — elevated in allergic/type 2-driven asthma
Early biomarker assessment enables physicians to identify patients at highest risk of remodeling and to select treatment strategies that target the root cause of disease progression.7
Clinical action |
Identify your patients at risk of airway remodeling — assess type 2 inflammation biomarkers early. Proactive phenotyping and targeted intervention can improve the trajectory of disease progression.7 |
Listen to Prof. Alberto Papi about the importance of early intervention
-
Nakagome K, Nagata M. The Possible Roles of IL-4/IL-13 in the Development of Eosinophil-Predominant Severe Asthma. Biomolecules. 2024;14(5):546. Published 2024 May 2. doi:10.3390/biom14050546
-
Scott G, Asrat S, Allinne J, et al. IL-4 and IL-13, not eosinophils, drive type 2 airway inflammation, remodeling and lung function decline. Cytokine. 2023;162:156091. doi:10.1016/j.cyto.2022.156091
-
Bradding P, Porsbjerg C, Côté A, Dahlén SE, Hallstrand TS, Brightling CE. Airway hyperresponsiveness in asthma: The role of the epithelium. J Allergy Clin Immunol. 2024;153(5):1181-1193. doi:10.1016/j.jaci.2024.02.011
-
Hough KP, Curtiss ML, Blain TJ, et al. Airway Remodeling in Asthma. Front Med (Lausanne). 2020;7:191. Published 2020 May 21. doi:10.3389/fmed.2020.00191
-
Chapman DG, Irvin CG. Mechanisms of airway hyper-responsiveness in asthma: the past, present and yet to come. Clin Exp Allergy. 2015;45(4):706-719. doi:10.1111/cea.12506
-
Abe Y, Suga Y, Fukushima K, et al. Advances and Challenges of Antibody Therapeutics for Severe Bronchial Asthma. Int J Mol Sci. 2021;23(1):83. Published 2021 Dec 22. doi:10.3390/ijms23010083
-
Zhang J, Dong L. Status and prospects: personalized treatment and biomarker for airway remodeling in asthma. J Thorac Dis. 2020;12(10):6090-6101. doi:10.21037/jtd-20-1024
-
León B, Ballesteros-Tato A. Modulating Th2 Cell Immunity for the Treatment of Asthma. Front Immunol. 2021;12:637948. Published 2021 Feb 10. doi:10.3389/fimmu.2021.637948
-
Pelaia C, Heffler E, Crimi C, et al. Interleukins 4 and 13 in Asthma: Key Pathophysiologic Cytokines and Druggable Molecular Targets. Front Pharmacol. 2022;13:851940. Published 2022 Mar 8. doi:10.3389/fphar.2022.851940
-
Gandhi NA, et al. Nat Rev Drug Discov. 2016;15(1):35-50.
-
Israel E, Reddel HK. N Engl J Med. 2017;377(3):965-976.
-
Mauad T, et al. J Allergy Clin Immunol. 2007;120(5):997-1009.
-
Robinson D, et al. Clin Exp Allergy. 2017;47(2):161-175.