Studier på: Astma

Registreringsstudier på svær astma og Dupixent
.png/jcr:content.png)
TRAVERSE
3 års opfølgningsstudie fra LIBERTY
Long-Term Efficacy of Dupilumab in Moderate-to-Severe Asthma Phenotyped by Blood Eosinophils and Exhaled Nitric Oxide
Michael E. Wechsler, MD; Ian D. Pavord, MD; Alberto Papi, MD; Kenneth R. Chapman, MD; Arman Altincatal, MS; Nami Pandit-Abid, PharmD; Juby A. Jacob-Nara, MD; Paul J. Rowe, MD; Yamo Deniz, MD; Elizabeth Laws, PhD; Bolanle Akinlade, MD; Nikhil Amin, MD; Heribert W. Staudinger, MD; David J. Lederer, MD; Megan Hardin, MD
.png/jcr:content.png)
EXCURSION
2 års opfølgningsstudie
Dupilumab 200 mg was efficacious in children (6–11 years) with moderate‐to‐severe asthma for up to 2 years: EXCURSION open‐label extension study
Wanda Phipatanakul MD, Christian Vogelberg MD, Leonard B. Bacharier MD, Sharon Dell MD, Arman Altincatal MS, Rebecca Gall MD, Oliver Ledanois MD, Harry Sacks MD, Juby A. Jacob‐Nara MD, Yamo Deniz MD, Paul J. Rowe MD

QUEST
Registreringsstudie fra LIBERTY
Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma.
Mario Castro, M.D., Jonathan Corren, M.D., Ian D. Pavord, M.D., et al: N Engl J Med 2018; 378:2486-2496. DOI: 10.1056/NEJMoa1804092

VENTURE
Registreringsstudie fra LIBERTY
Efficacy and Safety of Dupilumab in Glucocorticoid-Dependent Severe Asthma. Rabe KF, et al; N Engl J Med 2018; 378:2475-2485 DOI: 10.1056/NEJMoa1804093

TRAVERSE
3 års opfølgningsstudie fra LIBERTY
Long-term safety and efficacy of dupilumab in patients with moderate-to-severe asthma (TRAVERSE): an open-label extension study
Michael E Wechsler, Linda B Ford, Jorge F Maspero, Ian D Pavord, Alberto Papi, Arnaud Bourdin et al., Lancet Respir Med 2021, doi.org/10.1016/S2213-2600(21)00322-2

VOYAGE
Registreringsstudie - børn og svær astma - LIBERTY
Dupilumab in Children with Uncontrolled Moderate-to-Severe Asthma
L.B. Bacharier, J.F. Maspero, C.H. Katelaris, A.G. Fiocchi et al., N Engl J Med 2021;385:2230-40.
DOI: 10.1056/NEJMoa2106567

VOYAGE - EXCURSION
Assessment of long-term safety and efficacy of dupilumab in children with asthma (LIBERTY ASTHMA EXCURSION): an open-label extension study
Prof Leonard B Bacharier MD, Jorge F Maspero MD, Prof Constance H Katelaris MD, Alessandro G Fiocchi MD, Remi Gagnon MD, Ines de Mir MD, Prof Theresa W Guilbert MD, Prof Daniel J Jackson MD, Heribert W Staudinger MD, Elizabeth Laws PhD, Leda P Mannent MD, Bolanle Akinlade MD, Jennifer Maloney MD, Kelsey Tawo MD, Faisal A Khokhar MD, Ning Li MS, Megan Hardin MD, Raolat M Abdulai MD, David J Lederer MD, Lacey B Robinson MD, Lacey B. Robinson
Volume 12, Issue 1, January 2024, Pages 45-54 https://doi.org/10.1016/S2213-2600(23)0030-X
Sub-publikationer af registreringsstudier på svær astma og Dupixent
.jpg/jcr:content.jpg)
TRAVERSE - allergi
Dupilumab improves long-term outcomes in patients with uncontrolled, moderate-to-severe GINA-based type 2 asthma, irrespective of allergic status
Rabe KF, Pavord ID, Busse WW, Chupp GL, Izuhara K, Altincatal A, Gall R, Pandit-Abid N, Deniz Y, Rowe PJ, Jacob-Nara JA, Radwan A. Dupilumab improves long-term outcomes in patients with uncontrolled, moderate-to-severe GINA-based type 2 asthma, irrespective of allergic status. Allergy. 2023 Aug;78(8):2148-2156. doi: 10.1111/all.15747. Epub 2023 May 21. PMID: 37073882.

TRAVERSE - ICS
Dupilumab sustains efficacy in patients with moderate-to-severe type 2 asthma regardless of ICS dose
Pavord, I. D., Bourdin, A., Papi, A., Domingo, C., Corren, J., Altincatal, A., Radwan, A., Pandit-Abid, N., Jacob-Nara, J. A., Deniz, Y., Rowe, P. J., Laws, E., Lederer, D. J., & Hardin, M. (2023). Dupilumab sustains efficacy in patients with moderate-to-severe type 2 asthma regardless of ICS dose. Allergy, 10.1111/all.15792. Advance online publication. https://doi.org/10.1111/all.15792

TRAVERSE - EKSACERBATIONER
Impact of exacerbation history on long-term efficacy of dupilumab in patients with asthma
Corren J, Katelaris CH, Castro M, et al. Impact of exacerbation history on long-term efficacy of dupilumab in patients with asthma. ERJ Open Res 2023; in press (https://doi.org/10.1183/23120541.00037-2023).
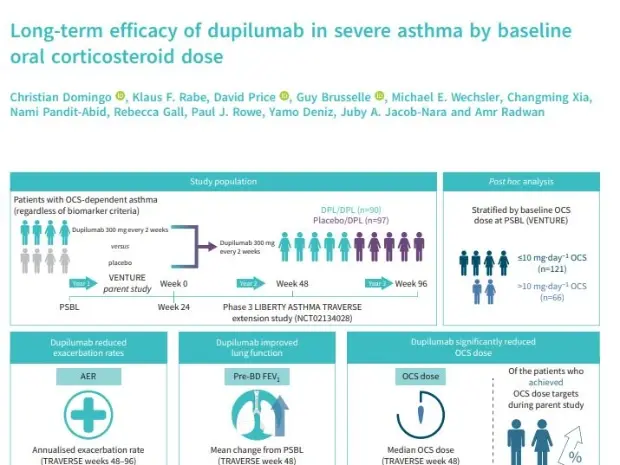
TRAVERSE - OCS
Long-term efficacy of dupilumab in severe asthma by baseline oral corticosteroid dose
Domingo C, Rabe KF, Price D, et al. Long-term efficacy of dupilumab in severe asthma by baseline oral corticosteroid dose. ERJ Open Res 2023; in press (https://doi.org/10.1183/23120541.00056-2023)

QUEST
Corren et al. - subpublikation af QUEST - allergi
Dupilumab Efficacy in Patients with Uncontrolled, Moderate-to-Severe Allergic Asthma - Corren J, Castro M, et al; J Allergy Clin Immunol Pract Volume 8, no. 2, February 2020;516-526

QUEST
Bourdin et al. - subpublikation af QUEST - højdosis ICS
Dupilumab is effective in type 2‐high asthma patients receiving high‐dose inhaled corticosteroids at baseline - Bourdin et al, Allergy 2021 Jan;76(1):269-280 doi: 10.1111/all.14611. Epub 2020 Oct 21.
.jpg/jcr:content.jpg)
QUEST - EKSACERBATIONER
Effect of exacerbation history on clinical response to dupilumab in moderate-severe uncontrolled asthma -
Corren J, Katelaris CH, Castro M, et al; Eur Respir J 2021

QUEST - HELÅRS ALLERIGSK RHINIT
Efficacy of dupilumab on clinical outcomes in patients with asthma and perennial allergic rhinitis
Busse et al, Ann Allergy Asthma Immunol Volume 125, Issue 5, November 2020, Pages 565-576

QUEST - BIOMARKØRER OG FALD I LUNGEFUNKTION
Biomarkers associated with lung function decline and dupilumab response in patients with asthma
Ian D. Pavord, Lucia de Prado Gomez, Guy Brusselle, Daniel J. Jackson, Christopher E. Brightling, Alberto Papi, Jorge F. Maspero, Klaus F. Rabe, Stephanie Korn, Mei Zhang, Xavier Soler, Juby A. Jacob-Nara, Megan Hardin, Nami Pandit-Abid, Amr Radwan, Yamo Deniz, Paul J. Rowe
MAT-DK-2400082(v1.0) 14. februar 2024
.webp/jcr:content.webp)
VOYAGE - ALLERGI
Assessment of dupilumab in children with moderate-to-severe type 2 asthma with or without evidence of allergic asthma
Papadopoulos NG, Szefler SJ, Bacharier LB, Maspero JF, Domingo C, Fiocchi A, Lee JK, Daizadeh N, Lederer DJ, Hardin M, Gall R, Djandji M, Siddiqui S, Jacob-Nara JA, Deniz Y, Rowe PJ. Assessment of dupilumab in children with moderate-to-severe type 2 asthma with or without evidence of allergic asthma. Allergy. 2023 Aug;78(8):2157-2167. doi: 10.1111/all.15743. Epub 2023 May 25. PMID: 37059696.

VOYAGE - QoL
Dupilumab leads to better-controlled asthma and quality of life in children: the VOYAGE study
Alessandro G. Fiocchi, Wanda Phipatanakul, Robert S. Zeiger, Sandy R. Durrani, Jeremy Cole, Jérôme Msihid, Rebecca Gall, Juby A. Jacob-Nara, Yamo Deniz, Paul J. Rowe, David J. Lederer, Megan Hardin, Yi Zhang, Asif H. Khan
European Respiratory Journal 2023 62: 2300558; DOI: 10.1183/13993003.00558-2023

VOYAGE - EOS / FeNO
Blood eosinophils and FeNO are prognostic and predictive biomarkers in childhood asthma
Leonard B. Bacharier, MD, Ian D. Pavord, MD, Jorge F. Maspero, MD, Daniel J. Jackson, MD, Alessandro G. Fiocchi, MD, Xuezhou Mao, PhD, Juby A. Jacob-Nara, MD, MPH, DHSc, Yamo Deniz, MD, Elizabeth Laws, PhD, Leda P. Mannent, MD, Nikhil Amin, MD, Bolanle Akinlade, MD, Heribert W. Staudinger, MD, PhD, David J. Lederer, MD, MS, Megan Hardin, MD, MPH
PII: S0091-6749(24)00043-5 DOI: https://doi.org/10.1016/j.jaci.2023.09.044

VOYAGE - LUNGEFUNKTION OG EKSACERBATIONER
Impact of Lung Function on Asthma Exacerbation Rates in Children Treated with Dupilumab: The VOYAGE Study
Theresa W Guilbert1, Kevin R Murphy2, Eckard Hamelmann3, Kristie R Ross4, Atul Gupta, Alessandro Fiocchi, Changming Xia, Rebecca Gall, Olivier Ledanois, Amr Radwan, Juby A Jacob-Nara, Paul J Rowe, Yamo Deniz. Journal of Asthma and Allergy, February 8, 2024. Doi: https://doi.org/10.2147/JAA.S425101
Andre relevante studier på svær astma og biologisk behandling

Reprint: Håkansson et al - OCS
Systemic corticosteroid use in type 2 inflammation-associated diseases including asthma, atopic dermatitis, allergic rhinitis, and chronic rhinosinusitis has been associated with adverse outcomes, and corticosteroid-sparing treatments are available.
Kjell Erik Julius Håkansson, Inge Raadal Skov, Steven Arild Wuyts Andersen, Zarqa Ali, Anders Løkke, Rikke Ibsen, Ole Hilberg, Howraman Meteran, Claus R Johnsen, Vibeke Backer, Charlotte Suppli Ulrik.

Reprint: biomarker and OCS – STAR 2025
Asthma characterization using blood eosinophil count (BEC) (among other biomarkers and clinical indices) is recommended in severe asthma (SA), but the masking effect of oral corticosteroids (OCS), makes this challenging.
Florence Schleich MD, PhD a, Désirée Larenas-Linnemann MD, FAAAAI, Dist.Intl.FACAAI b, Alan Altraja MD, PhD c, Luis Pérez de Llano MD, PhD d e, Konstantinos Kostikas MD, PhD, FERS f, Mohsen Sadatsafavi MD, PhD g, Arnaud Bourdin MD, PhD h, Roy Alton Pleasants PharmD i j, Mark Hew MBBS, PhD, FRACP k l, Wenjia Chen PhD m, Libardo Jiménez-Maldonado MD n o, Simon Couillard MD, MSc p, Charlotte Suppli Ulrik MD, DMSc q, Adeeb A. Bulkhi MD, MS r, Ming-Ju Tsai MD, PhD s t, George C. Christoff MD, MPH, PhD u, Nikolaos G. Papadopoulos MD, PhD, FRCP v w, Paul E. Pfeffer MRCP(UK), PhD x y, Dermot Ryan MD, FRCGP z, Celine Bergeron MD, FRCPC, MSc aa ab, Mona S. Al-Ahmad MD, FRCPC ac ad, Delbert R. Dorscheid MD, PhD ae, Eileen Wang MD, MPH af ag, John.

Reprint: OCS burden of disease, 2025
Over the past 70 years, oral corticosteroids (OCS) have played an important role in the management of acute and chronic asthma; however, their use is associated with an increased incidence of adverse events, chronic diseases such as osteoporosis and diabetes, and mortality, as well as increased healthcare resource utilization and costs. Despite a consensus that the use of OCS should be minimized in asthma treatment strategies, many patients still routinely receive long-term or frequent short courses of OCS. Add-on biologics can help to improve asthma control in patients with severe asthma and evidence of type 2 inflammation; in clinical trials and real-world studies, both short- and long-term OCS-sparing effects have been demonstrated. In this review, we discuss the benefits and burdens of OCS in patients with severe asthma from the perspective of both patients and healthcare providers and outline potential methods for reducing reliance on OCS. Given the risk of adrenal insufficiency or crisis upon cessation of OCS, we discuss the need for clear guidance and algorithms for the tapering of OCS together with the requirement for multidisciplinary patient–provider interactions as part of an effective OCS stewardship scheme.
G Walter Canonica, Celeste Porsbjerg, David B Price, Michael E Wechsler, Liam G Heaney, Nicola A Hanania, Rebecca Gall, Nami Pandit-Abid, Juby A Jacob-Nara, Harry J Sacks.

VESTIGE
Effect of dupilumab on exhaled nitric oxide, mucus plugs, and functional respiratory imaging in patients with type 2 asthma (VESTIGE): a randomised, double-blind, placebo-controlled, phase 4 trial
Castro M, Papi A, Porsbjerg C, Lugogo NL, Brightling CE, González-Barcala FJ, Bourdin A, Ostrovskyy M, Staevska M, Chou PC, Duca L, Pereira AM, Fogarty C, Nadama R, Zhang M, Rodrigues A, Soler X, Sacks HJ, Deniz Y, Rowe PJ, de Prado Gómez L, Jacob-Nara JA. Lancet Respir Med. 2025 Feb 10:S2213-2600(24)00362-X. doi: 10.1016/S2213-2600(24)00362-X. Epub ahead of print. PMID: 39947221.

VESTIGE – supplementary appendix
Supplement to: Castro M, Papi A, Porsbjerg C, et al. Effect of dupilumab on exhaled nitric oxide, mucus plugs, and functional respiratory imaging in patients with type 2 asthma (VESTIGE): a randomised, double-blind, placebo-controlled, phase 4 trial. Lancet Respir Med 2025; published online Feb 10. https://doi.org/10.1016/S2213-2600(24)00362-X.

NORDSTAR
Use of high-dose inhaled corticosteroids and risk of corticosteroid related adverse events in asthma -findings from the NORDSTAR cohort
Anna von Bülow, MD, PhD, Susanne Hansen, MSc, PhD, Patrik Sandin, PhD, Alexandra Cooper, MSc, Olivia Ernstsson, PhD, Kirk Geale, PhD, Lauri Lehtimäki, MD, PhD, Charlotte Ulrik, MD, DMsc, Bernt Bøgvald Aarli, MD, PhD, Pinja Ilmarinen, PhD, Sylvia Packham, PhD, Ghada Hassan, PhD, Asger Sverrild, MD, PhD, Helena Backman, PhD, Jussi Karjalainen, MD, Vibeke Backer, MD, DMsc, Alan Altraja, MD, PhD, Paula Kauppi, MD, PhD, Valentina Yasinska, MD, PhD, Maritta Kilpeläinen, MD, PhD, Arja Viinanen, MD, PhD, Johannes Martin-Schmid, MD, PhD, Apostolos Bossios, MD, PhD, Celeste Porsbjerg, MD, PhD, Hannu Kankaanranta, MD, PhD, Christer Janson, MD, PhD
.png/jcr:content.png)
RWE - Holland
Dupilumab responder types and predicting factors in patients with type 2 severe asthma: A real-world cohort study
L. Bult, J.C. Thelen, S.P. Rauh, J.C.C.M. in ‘t Veen, G.J. Braunstahl

SVÆR ASTMA OG BIOLOGISK BEHANDLING
Effective Management of Severe Asthma with Biologic Medications in Adult Patients: A Literature Review and International Expert Opinion
Roland Buhl MD, PhD, Elisabeth Bel MD, PhD, Arnaud Bourdin MD, PhD, Ignacio Dávila MD, PhD, Jo A. Douglass MD, J. Mark FitzGerald MD, David J. Jackson MRCP, PhD, Njira L. Lugogo MD, Andrea Matucci MD, Ian D. Pavord DM, Michael E. Wechsler MD, Monica Kraft MD
Volume 10, Issue 2, February 2022, Pages 422-432, https://doi.org/10.1016/j.jaip.2021.10.059

OVERBLIK - BIOLOGISK BEHANDLING
Biologics in severe asthma: A pragmatic approach for choosing the right
treatment for the right patient
Linda Rogers, Milos Jesenak, Leif Bjermer, Nicola A. Hanania, Sven F. Seys,
Zuzana Diamant
Respiratory Medicine 218 (2023) 107414 https://doi.org/10.1016/j.rmed.2023.107414

RWE - HOLLAND
Efficacy and safety of dupilumab as add-on therapy for patients with severe asthma: A real-world Dutch cohort study
Thelen, J. C., van Zelst, C. M., van Brummelen, S. E., Rauh, S., In 't Veen, J. C. C. M., Kappen, J. H., & Braunstahl, G. J. (2023). Efficacy and safety of dupilumab as add-on therapy for patients with severe asthma: A real-world Dutch cohort study. Respiratory medicine, 206, 107058. https://doi.org/10.1016/j.rmed.2022.107058
ORACLE
Predicting the benefits of type-2 targeted anti-inflammatory treatment with the prototype Oxford Asthma Attack Risk Scale (ORACLE)
Simon Couillard, William Il Hoon Do, Richard Beasley, Timothy S.C. Hinks, Ian D. Pavord
ERJ Open Research 2022 8: 00570-2021; DOI: 10.1183/23120541.00570-2021

Type 2 inflammation og lungefunktion
Type-2 inflammation and lung function decline in chronic airway disease in the general population
Yunus Çolak, Shoaib Afzal, Jacob Louis Marott, Jørgen Vestbo, Børge Grønne Nordestgaard, Peter Lange (2024). Type-2 inflammation and lung function decline in chronic airway disease in the general population.Thorax 2024;0:1–10. doi:10.1136/thorax-2023-220972
Klinisk remission studier - astma

Reprint: Håkansson et al – DSAR 2025
Severe asthma impacts work capacity, but little is known about the effects of biologic therapy on patients’ ability to work. We aimed to assess the impact of biologic therapy for severe asthma on work capacity and workforce attachment.
Kjell Erik Julius Håkansson, Rikke Ibsen, Niels Steen Krogh, Marianne Baastrup Soendergaard, Susanne Hansen, Anne-Sofie Bjerrum, Anna von Bülow, Ole Hilberg, Anders Løkke, Barbara Bonnesen, Claus Rikard Johnsen, Sofie Lock-Johansson, Lycely Dongo, Maria Bisgaard Borup, Roxana Vijdea, Linda Makowska Rasmussen, Johannes Martin Schmid, Charlotte Suppli Ulrik & Celeste Porsbjerg

Reprint: RWE DSAR economic consequences 2025
Severe asthma is seen in up to 5–10% of the total population with asthma, representing a significant portion of the asthma burden incurred by high healthcare resource utilisation and poor quality of life due to frequent exacerbations and loss of lung function.Citation1 The high burden of disease for individuals with severe asthma also affects both workplace attendance (absenteeism) and productivity (presenteeism).
Kjell Erik Julius Håkansson, Rikke Ibsen, Niels Steen Krogh, Marianne Baastrup Soendergaard, Susanne Hansen, Anne-Sofie Bjerrum, Anna von Bülow, Ole Hilberg, Anders Løkke, Barbara Bonnesen Bertelsen, Claus Rikard Johnsen, Sofie Lock Johansson, Lycely Dongo, Maria Bisgaard Borup, Roxana Vijdea, Linda Makowska Rasmussen, Johannes Martin Schmid, Charlotte Suppli Ulrik & Celeste Porsbjerg

Reprint: Hansen et al – Dupi and remission DSAR
Randomized clinical trials have demonstrated that dupilumab reduces exacerbations and maintenance oral corticosteroids (mOCS) use in patients with uncontrolled and severe asthma. However, evidence in real-life settings is limited.
Susanne Hansen, Kjell Erik Julius Håkansson, Marianne Baastrup Soendergaard, Anne-Sofie Bjerrum, Johannes Martin Schmid, Sofie Lock Johansson, Linda Makowska Rasmussen, Claus Rikard Johnsen, Anna von Bülow, Barbara Bonnesen, Niels Steen Krogh, Ole Hilberg, Lycely Dongo, Roxana Vijdea, Charlotte Suppli Ulrik, Celeste Porsbjerg.

RWE - ITALIEN
Severe asthma clinical remission after biologic treatment with anti-IL4/
IL13: A real-life experience
Andrea Portacci , Ilaria Iorillo, Vitaliano Nicola Quaranta, Leonardo Maselli, Ernesto Lulaj, Enrico Buonamico, Silvano Dragonieri,Giovanna Elisiana Carpagnano
Respiratory Medicine 217 (2023) 107348. https://doi.org/10.1016/j.rmed.2023.107348

RWE - DSAR DK
Clinical Response and Remission in Patients With Severe Asthma Treated With Biologic Therapies
Susanne Hansen, Marianne Søndergaard, Anna von Bülow, Anne-Sofie Bjerrum, Johannes Schmid, Linda M. Rasmussen, Claus R. Johnsen, Truls Ingebrigtsen, Kjell Erik Julius Håkansson, Sofie Lock Johansson, Maria Bisgaard, Karin Dahl Assing, Ole Hilberg, Charlotte Ulrik, Celeste Porsbjerg
Chestjournal.org, Available online 3 November 2023, https://doi.org/10.1016/j.chest.2023.10.046
Her finder du appendix til studierne:
- Supplementary - QUEST studiet - Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. Castro M, Corren J, Pavord ID, et al.N Engl J Med 2018;378:2486-96. DOI: 10.1056/NEJMoa1804092
- Supplementary - VENTURE studiet - Efficacy and safety of dupilumab in glucocorticoid-dependent severe asthma. Rabe KF, Nair P, Brusselle G, et al. N Engl J Med 2018;378:2475-85. DOI: 10.1056/NEJMoa1804093
For at læse denne side skal du være logget ind. Hvis du ikke har en konto, kan du oprette en her.
Præference center
Sundhedsperson verificering
Din konto bliver gennemgået og valideret. Du modtager en email, når din konto er blevet godkendt.
Gensendelse af email lykkedes
Gensend vellykket, tjek venligst din indbakke og klik på knappen inde for at bekræfte din e-mailadresse.
Unauthorized login
Leave site modal
Du er ved at forlade campus.sanofi/dk. Sanofi er ikke ansvarlig for indholdet af dette eksterne websted.
Arbejder du inden for sundhedsvæsenet?
Arbejder du inden for sundhedsvæsenet?
Denne hjemmeside indeholder kun information, der er rettet mod sundhedspersoner. Bekræft venligst nedenfor for at modtage information, film, podcasts og andet materiale.
