Hand och fot atopisk dermatit
Living with atopic dermatitis on your most essential body areas like the hands and feet can make daily activities including walking and writing incredibly burdensome even in the case where disease symptoms are mild elsewhere. Treating atopic dermatitis on the hands and feet has historically been difficult to treat.*1-3
Having Dupixent data added for this difficult-to-treat population is important for physicians looking for
tools to treat these patients and reinforces the already well-established efficacy and safety of Dupixent® (dupilumab) in atopic dermatitis overall.1,2
Dupixent®(dupilumab) in patients with atopic hand and foot dermatitis1-2
The Phase 3 double-blind, placebo-controlled trial, LIBERTY-AD-HAFT, evaluated the efficacy and safety of Dupixent in 133 adult and adolescent (aged 12 to 17 years) patients with atopic dermatitis with moderate-to-severe hand and/or foot involvement who had an inadequate response or intolerance to topical corticosteroids. Patients with hand and foot disease predominantly driven by allergic or irritant contact dermatitis were excluded from the trial.
The primary endpoint evaluated the proportion of patients with clear or almost clear skin of hand and feet eczema at 16 weeks, as measured by a score of 0 or 1 on the Investigator Global Assessment Scale. The key secondary endpoint measured the proportion of patients with improvement in itch on hands and feet from baseline (measured by a ≥4-point reduction in Peak-Pruritis Numeric Rating Scale [PP-NRS] on a 0-10 scale) at 16 weeks.
Proportion of patients achieving hand and foot IGA 0/1 over time2
Baseline: IGA of hand and foot (0–4), n: IGA 3 (moderate) placebo 48% (n=66) dupilumab 48 (N=67),
Patient was considered as non-responder after rescue treatment use or missing at each visit.
Proportion of patients achieving a 4-point improvement in weekly average of daily hand and foot PP-NRS over time**2
Baseline: Hand and foot PP-NRS (0–10), mean (SD)a, placebo 6.9 (n=66) dupilumab 7.2 (n=67)
Patient was considered as non-responder after rescue treatment use or missing at each visit.
The Phase 3 double-blind, placebo-controlled trial, LIBERTY-AD-HAFT, evaluated the efficacy and safety of Dupixent in 133 adult and adolescent (aged 12 to 17 years) patients with atopic dermatitis with moderate-to-severe hand and/or foot involvement who had an inadequate response or intolerance to topical corticosteroids. Patients with hand and foot disease predominantly driven by allergic or irritant contact dermatitis were excluded from the trial.
The primary endpoint evaluated the proportion of patients with clear or almost clear skin of hand and feet eczema at 16 weeks, as measured by a score of 0 or 1 on the Investigator Global Assessment Scale. The key secondary endpoint measured the proportion of patients with improvement in itch on hands and feet from baseline (measured by a ≥4-point reduction in Peak-Pruritis Numeric Rating Scale [PP-NRS] on a 0-10 scale) at 16 weeks.
Table 1. Patient baseline demographics2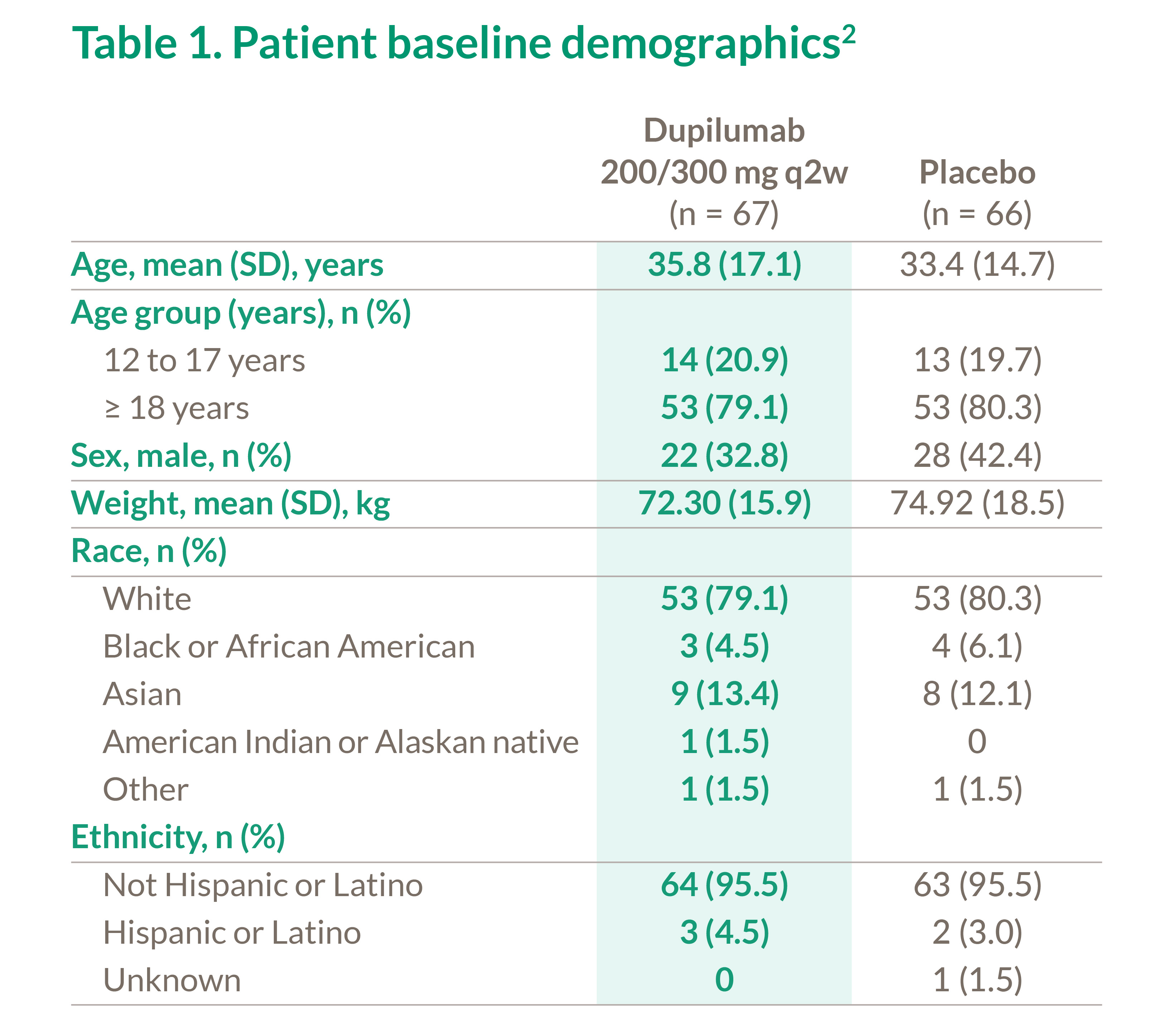
Table 2. Patient baseline disease characteristics2
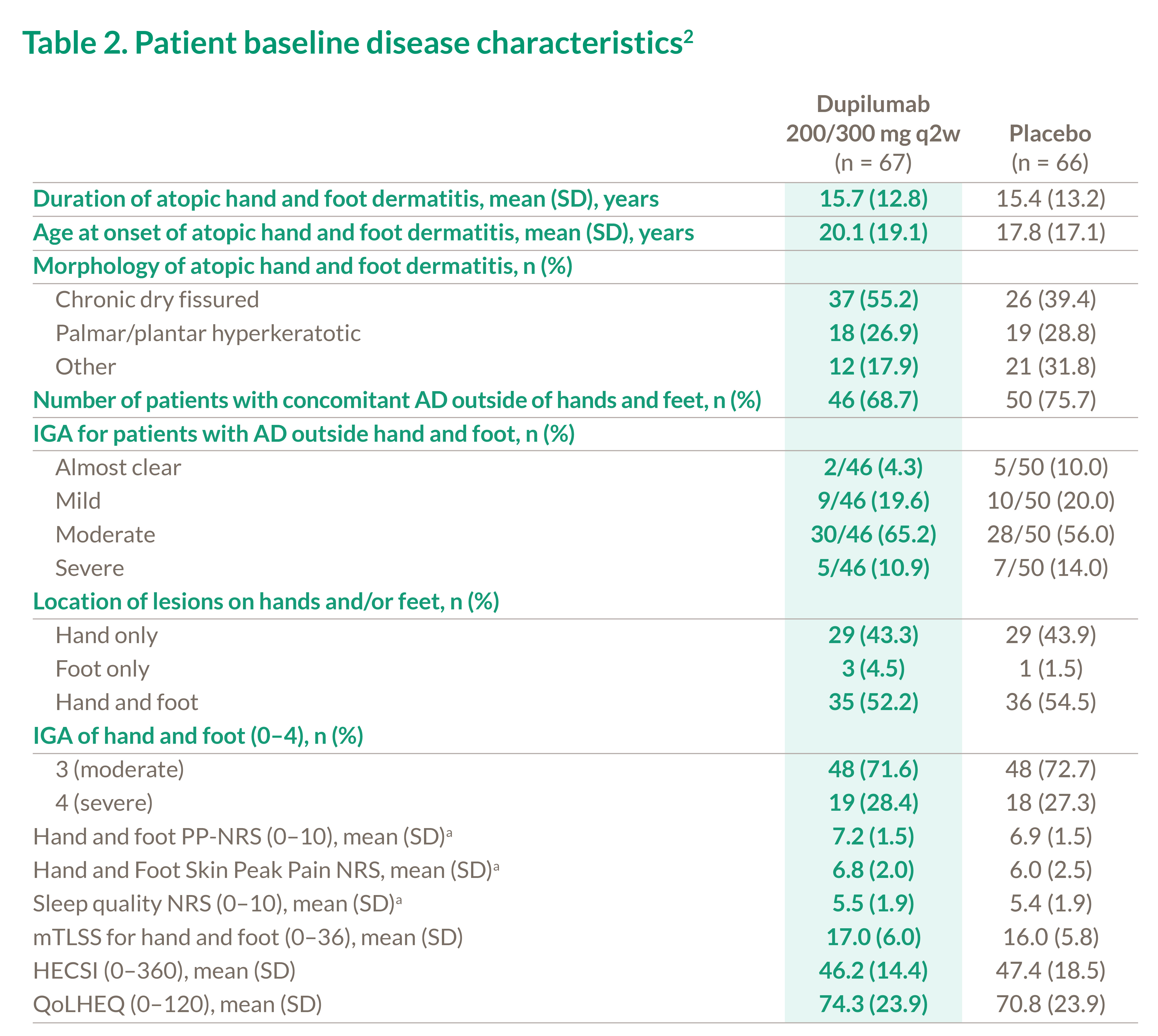
Figure 4. LS mean change in secondary PRO endpoints at Week 162
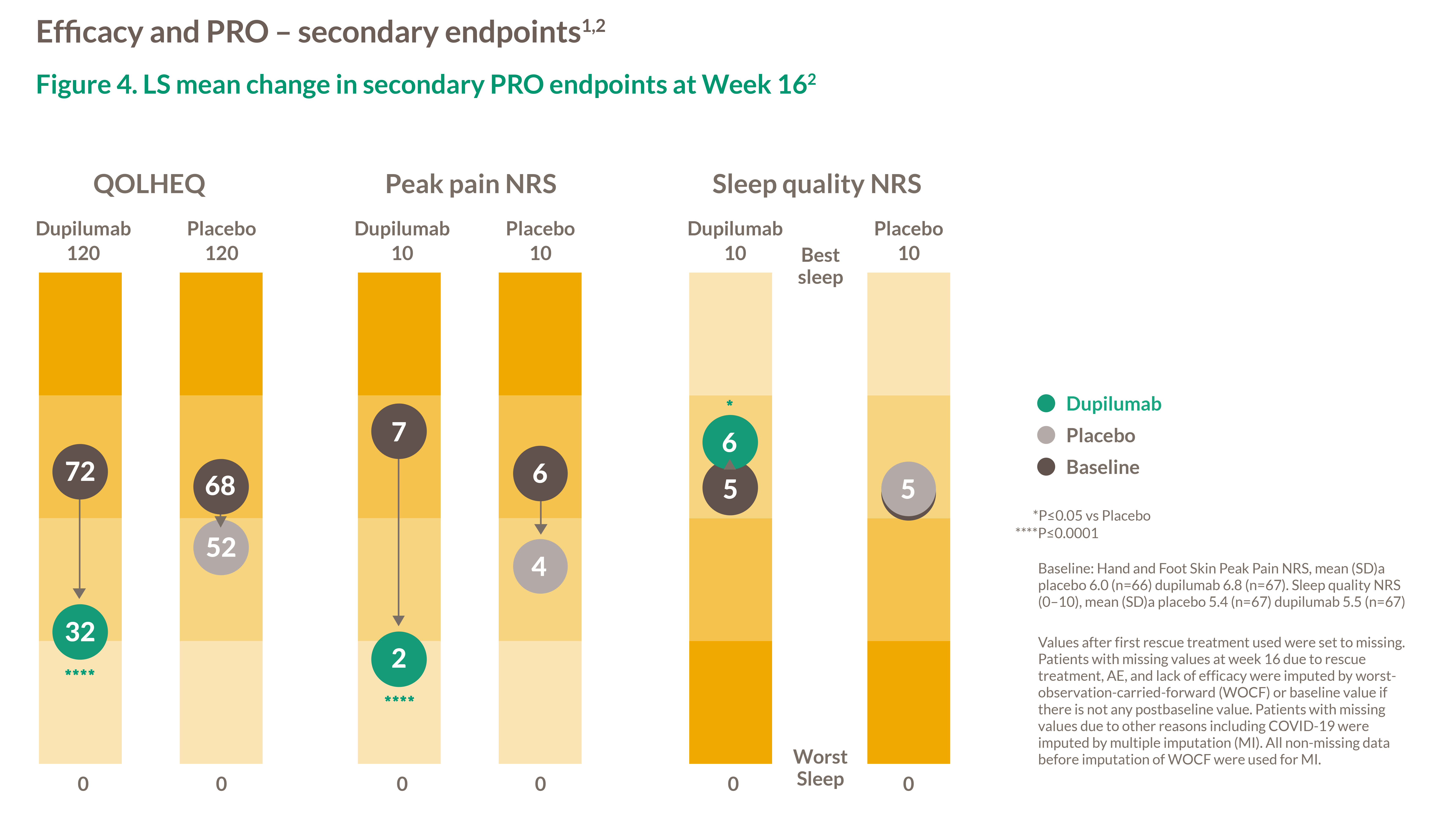
Baseline: Hand and Foot Skin Peak Pain NRS, mean (SD)a placebo 6.0 (n=66) dupilumab 6.8 (n=67). Sleep quality NRS (0–10), mean (SD)a placebo 5.4 (n=67) upilumab 5.5 (n=67)
Values after first rescue treatment used were set to missing. Patients with missing values at week 16 due to rescue treatment, AE, and lack of efficacy were imputed by worstobservation-carried-forward (WOCF) or baseline value if there is not any postbaseline value. Patients with missing values due to other reasons including COVID-19 were imputed by multiple imputation (MI). All non-missing data before imputation of WOCF were used for MI.
Dupixent safety profile was acceptable and was consistent with the known safety profile in the approved ad indication1,2
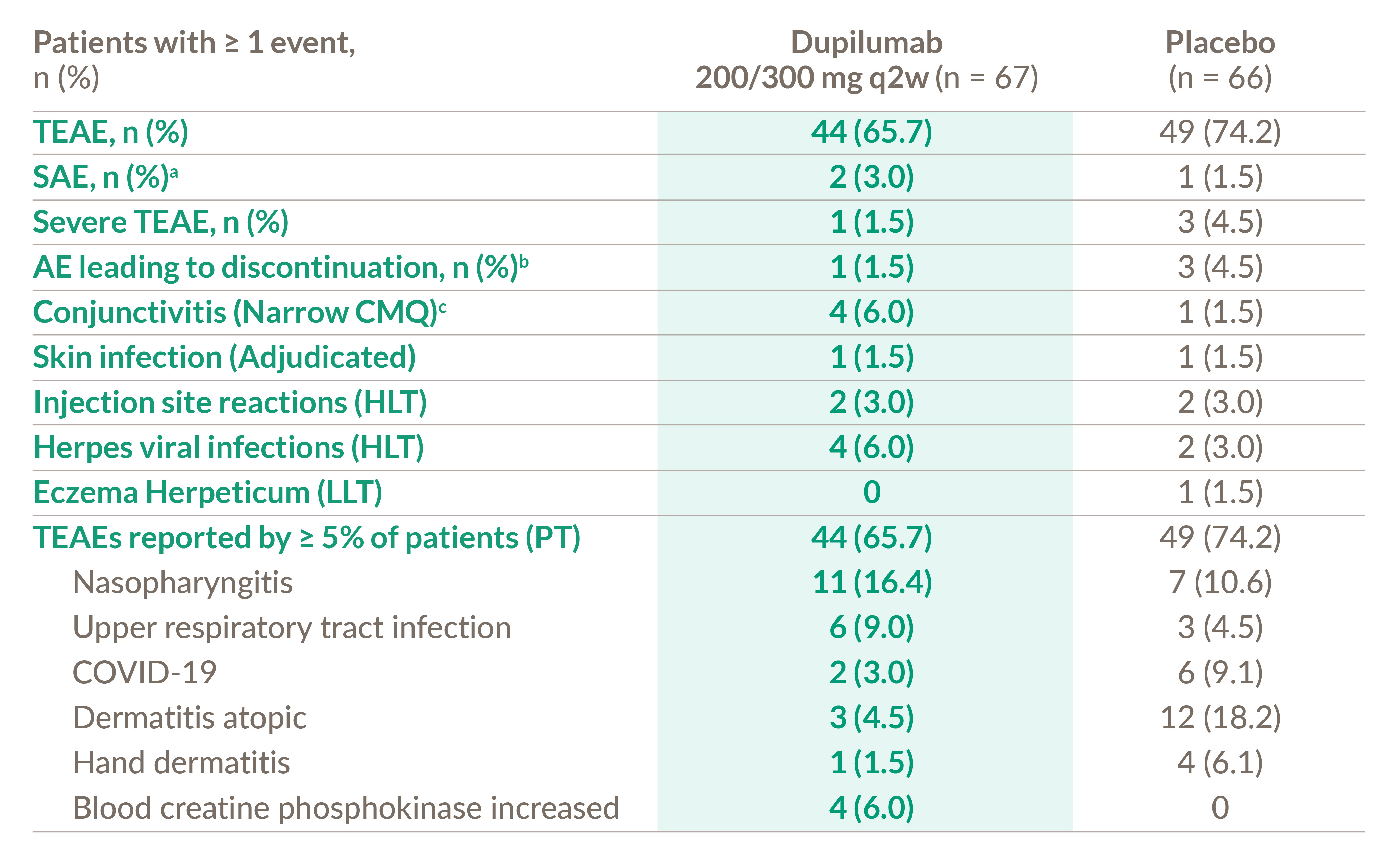 a) 1 patient reported appendicitis in the placebo group and 2 patients reported 3 events of post procedural infection, dizziness, and syncope in the dupilumab group.
a) 1 patient reported appendicitis in the placebo group and 2 patients reported 3 events of post procedural infection, dizziness, and syncope in the dupilumab group.
b) 3 patients reported AEs of general physical health deterioration, COVID-19, and dermatitis atopic leading to permanent study drug discontinuation in the placebo
group and 1 patient reported AE of dizziness and syncope that lead to permanent study drug discontinuation.
c) Narrow CMQ includes PTs of Conjunctivitis, Conjunctivitis allergic, Conjunctivitis bacterial, Conjunctivitis viral, Atopic keratoconjunctivitis.
Read the published article by clicking the link:
*DUPIXENT has demonstrated a significant improvement in effect and a long-term safety profile was observed up to 52 weeks in audlt patients with moderat to severe atopic dermatitis across different anatomical regions (post‐hoc analysis).3 DUPIXENT has also demonstrated a significant improvement in effect and a long-term safety profile observed up to 5 years in adult patients with moderate to severe AD.1Sustained significant improvement in effect and a long-term safety profile was observed up to 52 weeks in patients with severe AD from 6 years to 17 years.1Sustained significant improvement in effect and a safety profile was observed up to 16 weeks in patients with severe AD from 6 month to 5 years.1
Tables and graphs adapted by Sanofi.
**Weekly mean of daily measure.
NRS, Numerical Rating Scale (weekly mean of daily measure). PRO, patient reported outcomes; QoLHEQ, quality of life in hand eczema questionnaire (patients with hand dermatitis only).
CMQ, customized MeDRA queries; HLT, high level term; LLT, lowest level term; PT, preferred term;
SAE, serious adverse event; TEAE, treatment-emergent adverse event.
Ref: 1. Dupixent SmPC, fass.se. Ref: 2. Eric L. Simpson, et al. Dupilumab Treatment in Patients With Atopic Hand and Foot Dermatitis: Results From a Phase 3, Randomized, Double-Blind, Placebo-Controlled Trial, J AM ACAD DERMATOL, Published online February 23, 2024. Ref: 3 Blauvelt A et al. Br J Dermatol 2019;181(1):196-197
Vill du läsa mer om behandling med Dupixent för de olika indikationerna, klicka in på Fass.se


