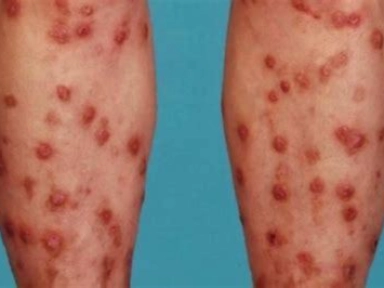
Efficacy and safety of dupilumab in patients with PN with or without a history of atopic comorbidities
Although the etiology of PN remains diverse and is not fully understood, a substantial proportion of patients with PN have concomitant atopic diseases: 50% AD, 31% asthma, and 71% allergic rhinitis (AR)1. In some patients, several diseases appear simultaneously, with almost 19% of patients with PN displaying the atopic triad (AD, asthma, and AR)1. However, in other patients, PN can exist without the presence of any atopic comorbidities.
While there is evidence that PN is a type 2 inflammatory disease distinct from AD, the efficacy of dupilumab in patients with PN with or without a history of atopic comorbidities has only been investigated recently both in a pre-specified subgroup analysis of the Phase III trials LIBERTY-PN PRIME and PRIME 22, and in long-term real-world evidence studies3.
This article summarizes evidence from this post-hoc analysis and recent real-world evidence studies from a French Early Access Program (EAP) and a long-term Italian retrospective study.
Key findings
- In randomized clinical trials, dupilumab significantly improved disease signs, symptoms, and health-related quality of life with similar onset time and response magnitude compared with placebo in adult patients with PN, irrespective of the presence or absence of atopic comorbidities2.
- Suppression of aberrant neuro-immune activity through the targeting of type 2 inflammation is suggested to have efficacy in the treatment of PN regardless of atopic background, IgE, or eosinophil levels2.
- In an Italian real-world retrospective study (104 weeks), the comparison between patients with or without concomitant atopic diathesis revealed similar clinical benefits obtained by dupilumab with a significantly more rapid reduction of IGA CPNs score and a significantly higher rate of patients achieving IGA CPNs of 0–1 in patients with concomitant atopic diathesis3.
- Patients that reached the last timepoint (104 weeks) validated some observations of delayed responses up to 1 year, demonstrating that complete resolution of PN may be achievable through long-term treatment3.
- Data from a French early access program (EAP, 12 months), predominantly including patients with non-atopic comorbidities or no comorbidities, shows that dupilumab progressively improves skin lesions and itch intensity over one-year period and demonstrates effectiveness regardless of atopic background4.
- Safety was generally consistent with the known dupilumab safety profile, both in randomized clinical trials and real-world settings2-4.
- These observations suggest that patients with PN may benefit from dupilumab treatment regardless of their atopic history2.
The objective of this study was to report the efficacy and safety of dupilumab in adult patients with moderate-to-severe PN with or without a history of atopic comorbidities, in a pre-specified subgroup analysis of pooled data from phase III trials LIBERTY-PN PRIME and PRIME2.
From 311 adult patients included, 43% of patients had a history or current atopic comorbidity (placebo n=68; dupilumab n=67), and 57% did not (placebo n=90; dupilumab n=86). Atopic and non-atopic subgroups were balanced between the dupilumab and placebo treatment arms, within each subgroup.
Within the atopic subgroup:
- 17.8% of patients had a history of atopic disease and 82.2% had an ongoing atopic disease.
- 9.6% had active mild AD (per protocol limited to 10%)
- 38.5% had two or more atopic comorbidities
- Allergic rhinitis was reported in 55.6% (51.9% ongoing)
- Asthma was reported in 33.3% (28.9% ongoing).
Efficacy Results: Signs and Symptoms Improvement at Week 24
Significantly more patients in the dupilumab versus the placebo group achieved clinically meaningful improvements across both subgroups at Week 24:
Table 1 Efficacy of Dupilumab on Itch and Skin Lesions at Week 24. The table is reproduced by Sanofi based on reference 2, figure 1.
Outcome at Week 24 |
Atopic (Dupilumab vs. placebo) proportion of patients (%)(placebo n = 68; dupilumab n = 67) |
Non-Atopic (Dupilumab vs. placebo) proportion of patients (%)(placebo n = 90; dupilumab n = 86) |
| Itch Reduction (Worst Itch Numeric Rating Scale (WI-NRS) improvement ≥ 4) |
58.2 vs 20.6 (P < 0.0001) |
59.3 vs 17.8 (P < 0.0001) |
| Clear/Almost Clear Skin (IGA PN-S 0 or 1) |
52.2 vs 16.2 (P < 0.0001) |
41.9 vs 17.8 (P = 0.0005) |
| Combined WI-NRS improvement ≥ 4 and Investigator’s Global Assessment (IGA) score of 0 or 1 |
37.3 vs 7.4 (P = 0.0057) |
33.7 vs 10.0 (P = 0.007) |
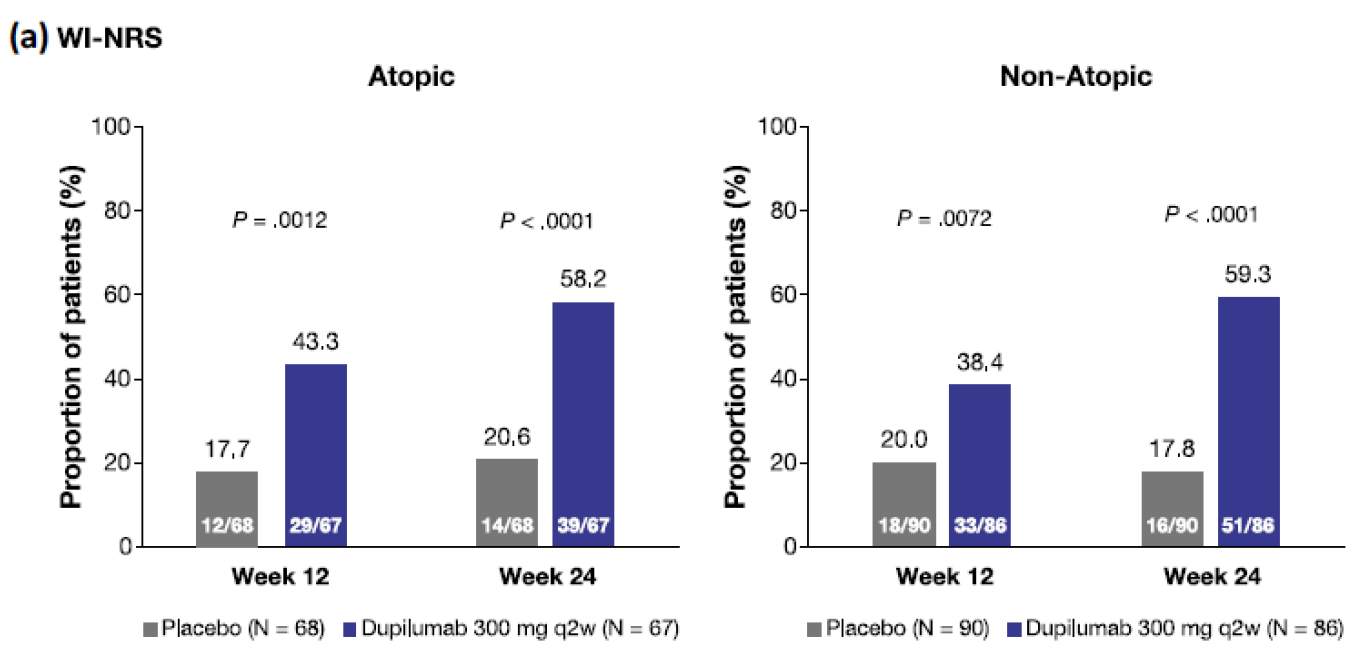
Figure 1 Efficacy of dupilumab on itch and skin lesions at week 12 and week 24 in patients with prurigo nodularis by atopic versus non-atopic subgroup. a Worst Itch Numeric Rating Scale (WI-NRS) improvement ≥ 4. The figure is copied by Sanofi based on reference 2, figure1.
Efficacy on other patient-reported outcomes at Week 24
The percent change from baseline in WI-NRS was similar at week 24 between dupilumab-treated patients in both subgroups. The onset of statistically significant improvement compared with placebo-treated patients was at week 3 in the atopic subgroup and at week 4 in the non-atopic subgroup.
Improvements in Skin Pain NRS, Dermatology Life Quality Index (DLQI) and Hospital Anxiety and Depression Scale (HADS) were similar in dupilumab-treated patients in both subgroups.
Table 2 Efficacy on other patient reported outcomes at week 24 by atopic versus non-atopic subgroup. The table is reproduced by Sanofi based on reference 2, figure 2.
Other patient reported outcomes at week 24 |
Atopic (Dupilumab vs. placebo) percentage change from baseline(placebo n = 68; dupilumab n = 67) |
Non-Atopic (Dupilumab vs. placebo) percentage change from baseline(placebo n = 90; dupilumab n = 86) |
| WI-NRS [0-10] |
-52.8 vs -22.7 (P < 0.0001) |
-57.0 vs -33.3% (P < 0.0001) |
| Skin Pain-NRS [0-10] |
-4.6 vs -2.5 (P < 0.0001) |
-4.4 vs -2.6 (P < 0.0001) |
| DLQI [0-30] |
-12.4 vs -5.8 (P < 0.0001) |
-12.9 vs -6.7 (P < 0.0001) |
| HADS [0-42] |
-3.8 vs -1.1 (P < 0.005) |
-6.0 vs -3.5 (P < 0.005) |
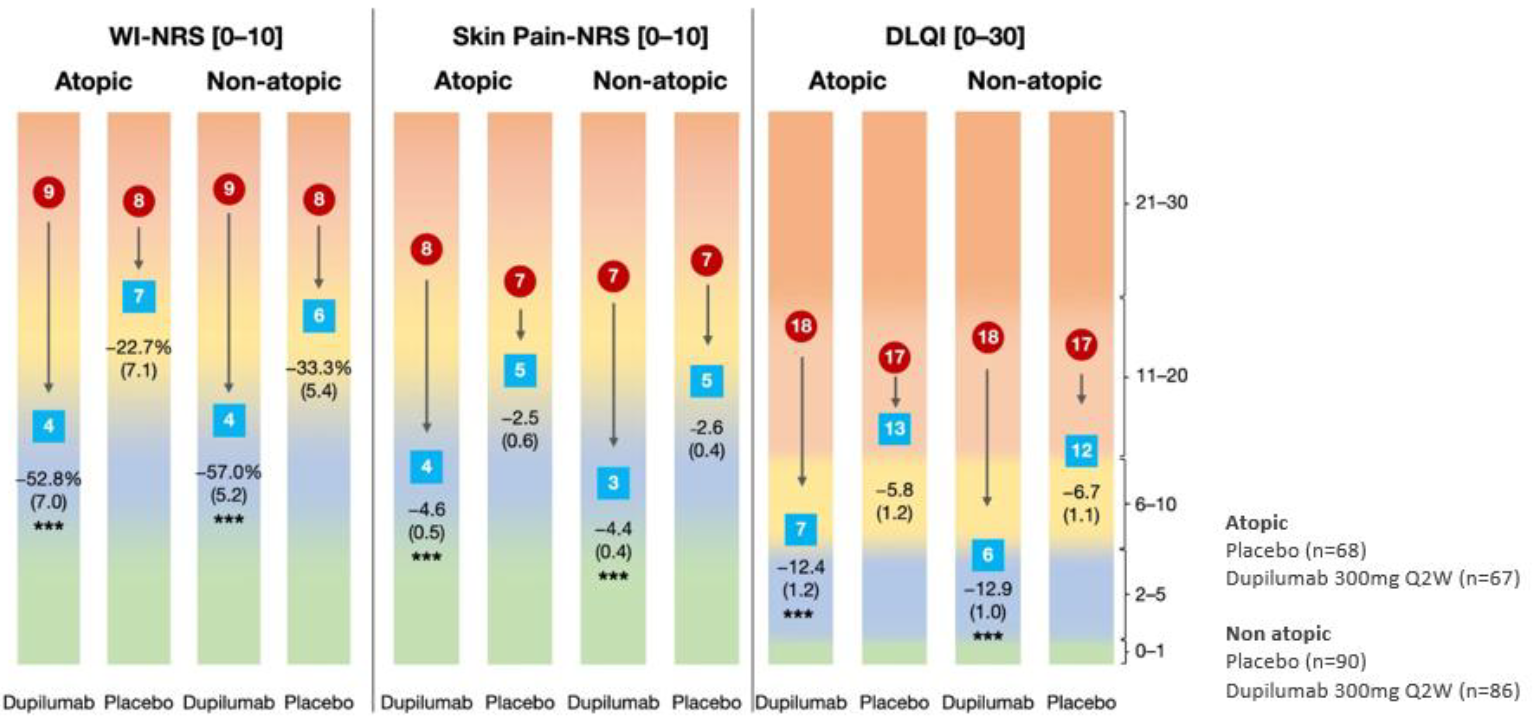
Figure 2 Efficacy on other patient reported outcomes at week 24 by atopic versus non-atopic subgroup. Efficacy on other patient-reported outcomes at week 24 by atopic versus non-atopic subgroup. Least squares mean percent change standard error). *P < 0.05 vs placebo; **P < 0.001 versus placebo; ***P < 0.0001 versus placebo. Scales: Worst Itch Numeric Rating Scale (WI-NRS): 0, “no itch” to 10, “worst imaginable itch”; Skin Pain-Numeric Rating Scale (Skin Pain-NRS): 0, “no pain” to 10, “worst imaginable pain”; Dermatology Life Quality Index (DLQI): 0, “no effect on patient’s life” to 30, “extremely large effect on patient’s life”; Hospital Anxiety and Depression Scale (HADS): two subscales, one for anxiety [0–21] and one for depression [0–21]: 0, “normal or no anxiety/depression” to 21, “severe anxiety/depression”. The figure is copied by Sanofi based on reference 2, figure 2.
Efficacy of Dupilumab in Patients with PN with Differing Baseline IgE and EoS Levels
At baseline, median serum total IgE levels were higher in atopic patients, however, total IgE levels were not informative in establishing a distinction between atopic and non-atopic patients in this study. Dupilumab treatment reduced total IgE levels in both subgroups, suggesting an effect on IgE regardless of the atopic status in PN. Furthermore, dupilumab treatment demonstrated a clinically meaningful improvement in itch, regardless of baseline total serum IgE and EoS levels, as well as an improvement in anxiety, depression, and quality of life, regardless of atopic status.
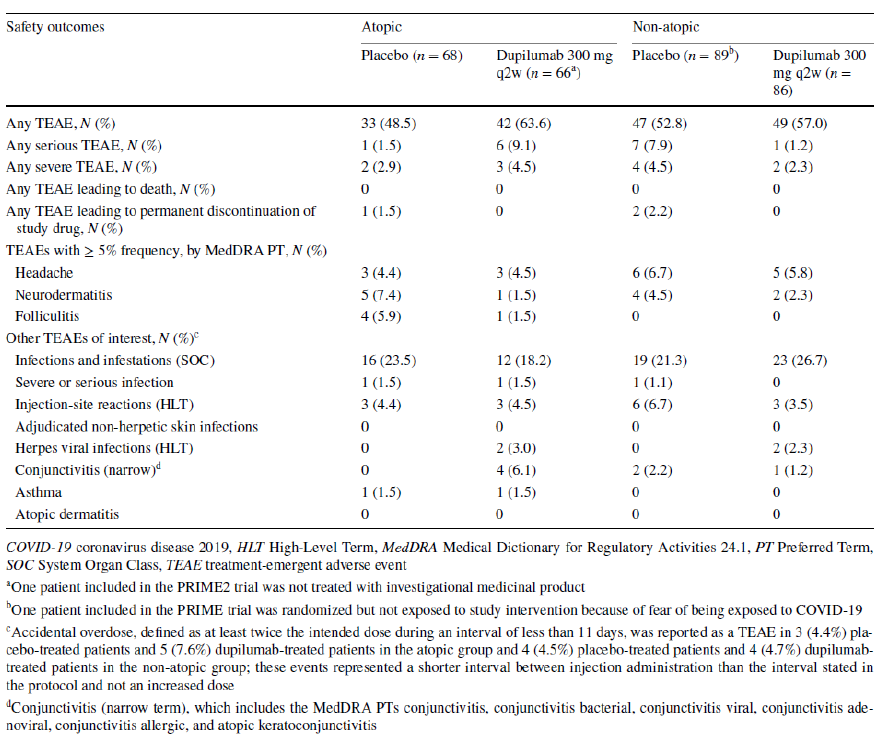
Overall safety profile across subgroups
The overall safety was generally consistent with the known dupilumab safety profile, with no remarkable differences across the subgroups. Treatment-emergent adverse events were reported in 63.6% (atopic) and 57.0% (non atopic) of dupilumab-treated patients, and there was no meaningful trend in serious TEAEs for atopic versus non-atopic patients.
Table 3 Summary of safety outcomes in the atopic and non-atopic patient subgroups. The table is copied by Sanofi based on reference 2, table 3.
Real-world evidence (RWE) is essential to complement randomized controlled trials (RCTs). Although RCTs are important for assessing efficacy, their patient populations and outcomes often differ from those seen in daily clinical practice. RWE helps us understand long-term effectiveness and safety in broader, non-selected patient groups5,6.
Long-term therapeutic response to dupilumab in patients affected by prurigo nodularis: A real-world retrospective study up to 104 weeks from Italy3
An Italian retrospective study of 64 adult patients showed that complete resolution of PN could be achievable through long-term treatment.
- Prior to starting dupilumab, 75% of patients showed a high disease burden, with moderate-to-severe skin manifestations, severe itch, and excoriations. Patients also showed marked impairment of quality of life.
- By week 16 of dupilumab treatment, 85% of patients achieved itch-NRS reduction ≥ 4, increasing to 97.5% by week 32, and subsequent maintenance of the clinical response throughout the study.
- The clinical benefits were similar for patients with or without atopic diathesis, with a more rapid reduction of IGA CPNs score.
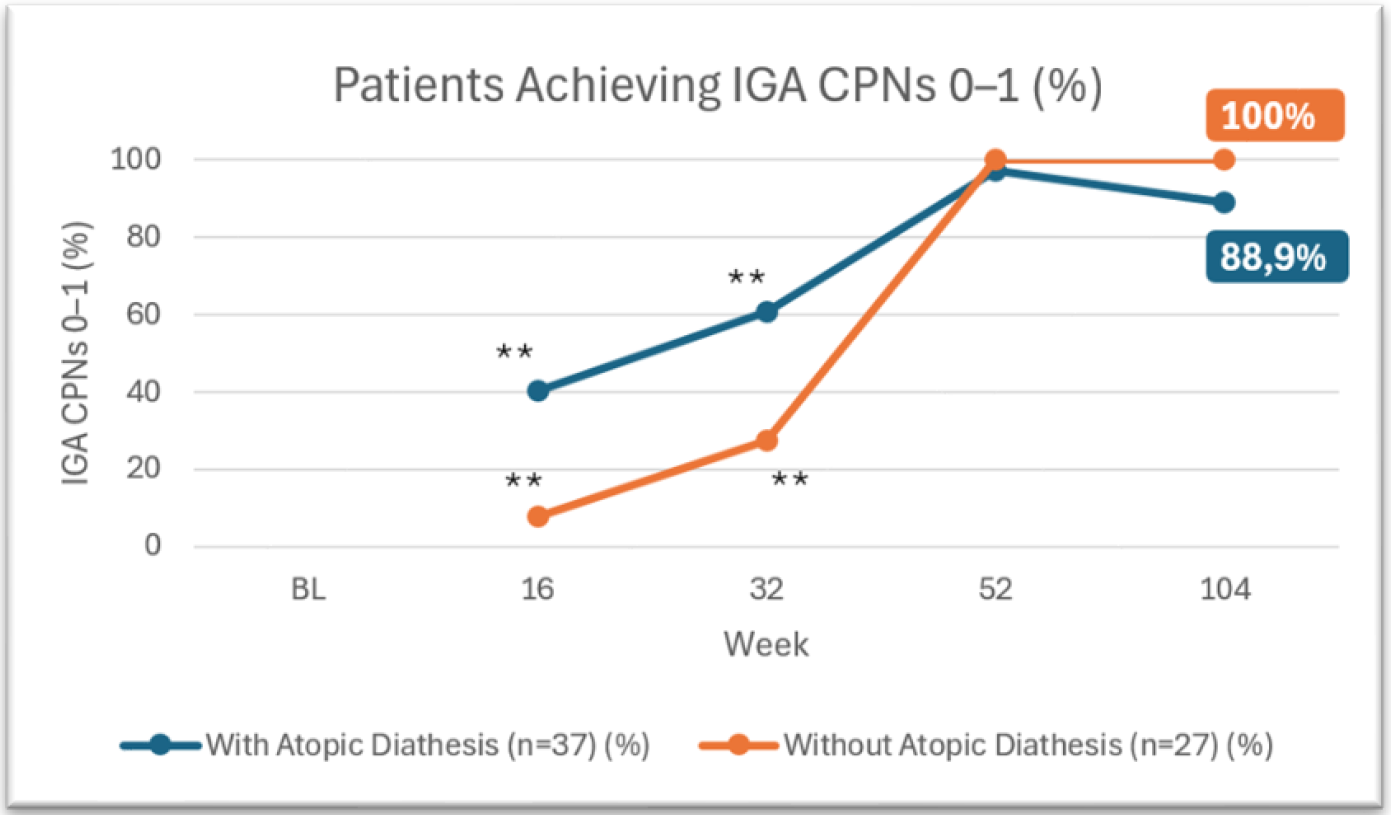
Figure 3 Patients achieving IGA CPNs 0-1. This figure is reproduced by Sanofi based on reference 3, table 2.
IGA CPNs (Investigator Global Assessment Chronic Prurigo Nodularis Staging): a 5-points physician reported outcome assessing the number of nodules (0 = Clear), no pruriginous lesions (0 lesions); 1 = almost clear, rare palpable pruriginous lesions (approximately 1–5 lesions); 2 = mild, few palpable pruriginous lesions (approximately 6–19 lesions); 3 moderate, many palpable pruriginous lesions (approximately 20–100 lesions); 4 = severe; abundant palpable pruriginous lesions (over 100 lesions).
**p < 0.05 for comparison between CPN patients with or without atopic diathesis.
- Nine total adverse events were reported during the observation period, with arthralgias being relatively more frequent; individual cases of conjunctivitis, injection site reactions, and oral candida were also noted.
The findings are consistent with those described in phase 3 trials but suggest a favourable effectiveness and safeness in the long-term period with a sustained and progressive amelioration of CPN manifestations beyond 24 weeks. The results also suggested a more rapid response in CPN patients with atopic diathesis that may likely reflect an enhanced type 2 inflammatory signal that could predominantly characterize the immune endotype of CPN, exceeding other immune signals.
Dupilumab in Adult Patients with Moderate-to-Severe PN: 12-Months Real-world Follow-up Results from the French Early Access Program4
In a cohort of 155 patients where 56.1% presented with only non-atopic comorbidities and 27.1% had no comorbidities, dupilumab demonstrated effectiveness regardless of atopic background. This analysis confirms progressive improvements over 1 year, with significant benefit observed as early as 12 weeks.
- Patients achieving clear or almost clear skin (IGA PN-S 0/1) increased over time, from 39.8% at 3 months to 82.2% at 12 months
- WI-NRS score reductions of ≥4 points were reported in 52.4% of patients at 3 months and reached 72.7% by 12 months
- The safety profile remained consistent with pivotal trials with no new safety concerns identified. The most common adverse events were headache and pruritus (1.9% each).
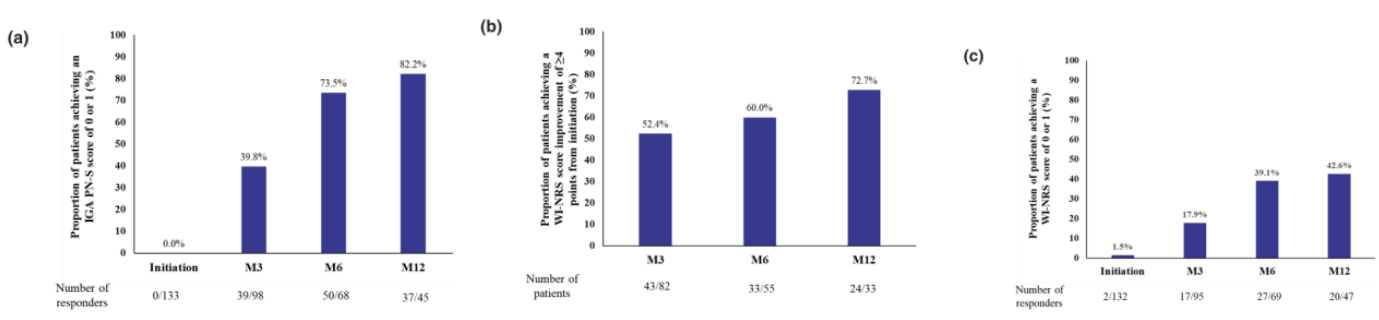
Figure 4 (a). Proportion of patients with PN with an IGA PN-S score of 0 or 1. (b) Proportion of patients with PN with an improvement of ≥4 points in the WI-NRS score. (c) Proportion of patients with PN with a WI-NRS score of 0 or 1. M3, M6 and M12 represent follow-up at 3, 6 and 12 months, respectively, after treatment initiation. IGA PN-S, Investigator's Global Assessment for Prurigo Nodularis-Stage; M, month; PN, prurigo nodularis; SE, standard error; WI-NRS, Worst-Itch Numerical Rating Scale. Figure copied by Sanofi based on the reference 4, figure 1.
Conclusion
Dupilumab significantly improved disease signs, symptoms, and health-related quality of life with similar onset time and response magnitude compared with placebo in adult patients with PN, irrespective of the presence or absence of atopic comorbidities. These benefits were sustained in the long-term, with real-world evidence showing high rates of skin clearance and itch relief through 1 year and beyond. These observations suggest that the suppression of aberrant neuro-immune activity through targeting type 2 inflammation has efficacy in treating PN regardless of atopic background, IgE, or eosinophil levels.
-
Aggarwal P, et al. Clin Exp Dermatol. 2021;46(7):1277–84.
-
Kim et al. Am J Clin Dermatol . 2025 Nov 11. Online ahead of print.
-
Chiricozzi A, et al. J Eur Acad Dermatol Venereol. 2024;38(10):e892-e895.
-
Jachiet M, et al. J Eur Acad Dermatol Venereol. Published online January 7, 2026.
-
Blonde L, et al. Adv Ther. 2018;35:1763–774
-
Heneghan C, et al. Trials. 2017;18:122